Febrile temperature change modulates CD4 T cell differentiation via a TRPV channel-regulated Notch-dependent pathway
- PMID: 32839313
- PMCID: PMC7486768
- DOI: 10.1073/pnas.1922683117
Febrile temperature change modulates CD4 T cell differentiation via a TRPV channel-regulated Notch-dependent pathway
Abstract
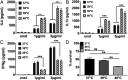
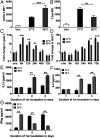
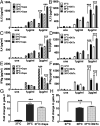
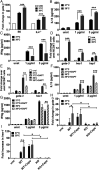
Fever is a conserved and prominent response to infection. Yet, the issue of how CD4 T cell responses are modulated if they occur at fever temperatures remains poorly addressed. We have examined the priming of naive CD4 T cells in vitro at fever temperatures, and we report notable fever-mediated modulation of their cytokine commitment. When naive CD4 T cells were primed by plate-bound anti-CD3 and anti-CD28 monoclonal antibodies at moderate fever temperature (39 °C), they enhanced commitment to IL4/5/13 (Th2) and away from IFNg (Th1). This was accompanied by up-regulation of the Th2-relevant transcription factor GATA3 and reduction in the Th1-relevant transcription factor Tbet. Fever sensing by CD4 T cells involved transient receptor potential vanilloid cation channels (TRPVs) since TRPV1/TRPV4 antagonism blocked the febrile Th2 switch, while TRPV1 agonists mediated a Th2 switch at 37 °C. The febrile Th2 switch was IL4 independent, but a γ-secretase inhibitor abrogated it, and it was not found in Notch1-null CD4 T cells, identifying the Notch pathway as a major mediator. However, when naive CD4 T cells were primed via antigen and dendritic cells (DCs) at fever temperatures, the Th2 switch was abrogated via increased production of IL12 from DCs at fever temperatures. Thus, immune cells directly sense fever temperatures with likely complex physiological consequences.
Keywords: Fever; Notch; TRPV; Th1/Th2.
Conflict of interest statement
Competing interest statement: S.R. is a nonexecutive director of Ahammune Biosciences Private Limited, Pune, India and a member of the scientific advisory boards of Curadev Pharma Private Limited, NOIDA, India, and Mynvax Private Limited, Bangalore, India. The other authors have no competing interests to declare.
Figures


References
-
- Schulman C. I., et al. , The effect of antipyretic therapy upon outcomes in critically ill patients: A randomized, prospective study. Surg. Infect. 6, 369–375 (2005). Correction in: Surg. Infect.11, 495 (2010). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

