Dexmedetomidine: What's New for Pediatrics? A Narrative Review
- PMID: 32846947
- PMCID: PMC7565844
- DOI: 10.3390/jcm9092724
Dexmedetomidine: What's New for Pediatrics? A Narrative Review
Abstract
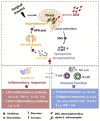
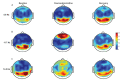
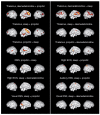
Over the past few years, despite the lack of approved pediatric labelling, dexmedetomidine's (DEX) use has become more prevalent in pediatric clinical practice as well as in research trials. Its respiratory-sparing effects and bioavailability by various routes are only some of the valued features of DEX. In recent years the potential organ-protective effects of DEX, with the possibility for preserving neurocognitive function, has put it in the forefront of clinical and bench research. This comprehensive review focused on the pediatric literature but presents relevant, supporting adult and animal studies in order to detail the recent growing body of literature around the pharmacology, end-organ effects, organ-protective effects, alternative routes of administration, synergetic effects, and clinical applications, with considerations for the future.
Keywords: anesthesia; neuroprotection; neurotoxicity; pediatric; pharmacology; sedation; side effects.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

References
-
- van Dijkman S.C., De Cock P., Smets K., Decaluwe W., Smits A., Allegaert K., Vande Walle J., De Paepe P., Della Pasqua O. Dose rationale and pharmacokinetics of dexmedetomidine in mechanically ventilated new-borns: Impact of design optimisation. Eur. J. Clin. Pharmacol. 2019;75:1393–1404. doi: 10.1007/s00228-019-02708-y. - DOI - PubMed
-
- Hannivoort L.N., Eleveld D.J., Proost J.H., Reyntjens K.M., Absalom A.R., Vereecke H.E., Struys M.M. Development of an Optimized Pharmacokinetic Model of Dexmedetomidine Using Target-controlled Infusion in Healthy Volunteers. Anesthesiology. 2015;123:357–367. doi: 10.1097/ALN.0000000000000740. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

