Plasmonic Nanoparticle-Based Digital Cytometry to Quantify MUC16 Binding on the Surface of Leukocytes in Ovarian Cancer
- PMID: 32847358
- PMCID: PMC7871419
- DOI: 10.1021/acssensors.0c00567
Plasmonic Nanoparticle-Based Digital Cytometry to Quantify MUC16 Binding on the Surface of Leukocytes in Ovarian Cancer
Abstract
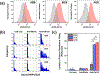
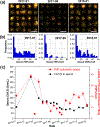
Although levels of the circulating ovarian cancer marker (CA125) can distinguish ovarian masses that are likely to be malignant and correlate with severity of disease, serum CA125 has not proved useful in general population screening. Recently, cell culture studies have indicated that MUC16 may bind to the Siglec-9 receptor on natural killer (NK) cells where it downregulates the cytotoxicity of NK cells, allowing ovarian cancer cells to evade immune surveillance. We present evidence that the presence of MUC16 can be locally visualized and imaged on the surface of peripheral blood mononuclear cells (PBMCs) in ovarian cancer via a novel "digital" cytometry technique that incorporates: (i) OC125 monoclonal antibody-conjugated gold nanoparticles as optical nanoprobes, (ii) a high contrast dark-field microscopy system to detect PBMC-bound gold nanoparticles, and (iii) a computational algorithm for automatic counting of these nanoparticles to estimate the quantity of surface-bound MUC16. The quantitative detection of our technique was successfully demonstrated by discriminating clones of the ovarian cancer cell line, OVCAR3, based on low, intermediate, and high expression levels of MUC16. Additionally, PBMC surface-bound MUC16 was tracked in an ovarian cancer patient over a 17 month period; the results suggest that the binding of MUC16 on the surface of immune cells may play an early indicator for recurrent metastasis 6 months before computational tomography-based clinical diagnosis. We also demonstrate that the levels of surface-bound MUC16 on PBMCs from five ovarian cancer patients were greater than those from five healthy controls.
Keywords: MUC16/CA125; computational analysis; dark-field microscopy; digital cytometry; leukocytes; longitudinal study; ovarian cancer; plasmonic gold nanoparticle.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

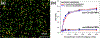
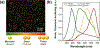
References
-
- Siegel RL; Miller KD; Jemal A Cancer statistics, 2018. Ca-Cancer J. Clin. 2018, 68, 7–30. - PubMed
-
- Naora H; Montell DJ Ovarian Cancer Metastasis: Integrating insights from disparate model organisms. Nat. Rev. Cancer 2005, 5, 355. - PubMed
-
- Rosen DG; Wang L; Atkinson JN; Yu Y; Lu KH; Diamandis EP; Hellstrom I; Mok SC; Liu J; Bast RC Jr. Potential markers that complement expression of CA125 in epithelial ovarian cancer. Gynecol. Oncol. 2005, 99, 267–277. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

