Efficacy of Combination Therapy With Pirfenidone and Low-Dose Cyclophosphamide for Refractory Interstitial Lung Disease Associated With Connective Tissue Disease: A Case-Series of Seven Patients
- PMID: 32851366
- PMCID: PMC7406158
- DOI: 10.46497/ArchRheumatol.2020.7381
Efficacy of Combination Therapy With Pirfenidone and Low-Dose Cyclophosphamide for Refractory Interstitial Lung Disease Associated With Connective Tissue Disease: A Case-Series of Seven Patients
Abstract
Objectives: This study reports a low dose combination therapy of cyclophosphamide (CYC) and pirfenidone (PFD) and the efficiency and safety of the therapy in refractory connective tissue disease associated interstitial lung disease (CTD-ILD) patients.
Patients and methods: The study included seven CTD-ILD patients (2 males, 5 females; mean age 48.8 years; range, 32 to 63 years) treated between January 2016 and December 2017 in our clinic. At enrolment, all patients had shown no improvement in their symptoms (dyspnea or cough) after at least one month of high dose steroids treatment. Patients who had received adjusted immunosuppressive agents other than steroids or anti-fibrotic medications within the three months before enrolment were excluded. We changed the treatment to a low dose combination of CYC 0.4 g/m2 monthly and PFD 300 mg twice per day and quickly reduced the steroids. All the patients were followed-up for 12 months.
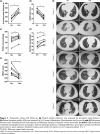
Results: Two patients had anti-synthetase syndrome, two had Sjögren syndrome, two had scleroderma and one had mixed connective tissue disease. The baseline forced vital capacity (FVC) was 39-81% and the six-minute walk distance (6MWD) was 202 m-324 m. Within 12 months follow-up, the median improvement in the FVC was 13.4% (range, 0-35.9%), the median improvement of carbon monoxide diffusing capacity was 6.3% (range, 1.7-16%) and the median improvement of 6MWD was 52.7% (range, 34.4-86.3%). All the patients were self-sufficient, and their dyspnea, chest high- resolution computed tomography scores, and quality of life improved simultaneously. Exceeding our expectations, no adverse events associated with CYC or PFD were observed during the follow-up period.
Conclusion: Our study provided preliminary while promising clinical evidence for combination therapy of CYC-PFD for CTD-ILD. A low dose combination of CYC and PFD was unexpectedly well tolerated, with satisfactory effects in refractory CTD-ILD patients. Well-designed controlled studies are needed to further establish the safety and efficacy of this approach.
Keywords: Cyclophosphamide; interstitial lung disease; pirfenidone.
Copyright © 2020, Turkish League Against Rheumatism.
Conflict of interest statement
Conflict of Interest: The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
Figures
References
-
- Yazisiz V, Arslan G, Ozbudak IH, Turker S, Erbasan F, Avci AB, et al. Lung involvement in patients with primary Sjögren’s syndrome: what are the predictors. Rheumatol Int. 2010;30:1317–1324. - PubMed
-
- Tashkin DP, Elashoff R, Clements PJ, Goldin J, Roth MD, Furst DE, et al. Cyclophosphamide versus placebo in scleroderma lung disease. N Engl J Med. 2006;354:2655–2666. - PubMed
LinkOut - more resources
Full Text Sources
Medical
