Accumulation of integrase strand transfer inhibitor resistance mutations confers high-level resistance to dolutegravir in non-B subtype HIV-1 strains from patients failing raltegravir in Uganda
- PMID: 32853364
- PMCID: PMC7662175
- DOI: 10.1093/jac/dkaa355
Accumulation of integrase strand transfer inhibitor resistance mutations confers high-level resistance to dolutegravir in non-B subtype HIV-1 strains from patients failing raltegravir in Uganda
Abstract
Background: Increasing first-line treatment failures in low- and middle-income countries (LMICs) have led to increased use of integrase strand transfer inhibitors (INSTIs) such as dolutegravir. However, HIV-1 susceptibility to INSTIs in LMICs, especially with previous raltegravir exposure, is poorly understood due to infrequent reporting of INSTI failures and testing for INSTI drug resistance mutations (DRMs).
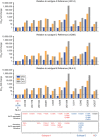
Methods: A total of 51 non-subtype B HIV-1 infected patients failing third-line (raltegravir-based) therapy in Uganda were initially selected for the study. DRMs were detected using Sanger and deep sequencing. HIV integrase genes of 13 patients were cloned and replication capacities (RCs) and phenotypic susceptibilities to dolutegravir, raltegravir and elvitegravir were determined with TZM-bl cells. Spearman's correlation coefficient was used to determine cross-resistance between INSTIs.
Results: INSTI DRMs were detected in 47% of patients. HIV integrase-recombinant virus carrying one primary INSTI DRM (N155H or Y143R/S) was susceptible to dolutegravir but highly resistant to raltegravir and elvitegravir (>50-fold change). Two patients, one with E138A/G140A/Q148R/G163R and one with E138K/G140A/S147G/Q148K, displayed the highest reported resistance to raltegravir, elvitegravir and even dolutegravir. The former multi-DRM virus had WT RC whereas the latter had lower RCs than WT.
Conclusions: In HIV-1 subtype A- and D-infected patients failing raltegravir and harbouring INSTI DRMs, there is high-level resistance to elvitegravir and raltegravir. More routine monitoring of INSTI treatment may be advised in LMICs, considering that multiple INSTI DRMs may have accumulated during prolonged exposure to raltegravir during virological failure, leading to high-level INSTI resistance, including dolutegravir resistance.
© The Author(s) 2020. Published by Oxford University Press on behalf of the British Society for Antimicrobial Chemotherapy. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures


References
-
- Raffi F, Rachlis A, Stellbrink H-J et al. Once-daily dolutegravir versus raltegravir in antiretroviral-naive adults with HIV-1 infection: 48 week results from the randomised, double-blind, non-inferiority SPRING-2 study. Lancet 2013; 381: 735–43. - PubMed
-
- Update of Recommendations on First-and Second-Line Antiretroviral Regimes, 2019. https://apps.who.int/iris/bitstream/handle/10665/325892/WHO-CDS-HIV-19.1....
-
- ARV Market Report: The State of the Antiretroviral Drug Market in Low- And Middle-Income Countries, 2016-2021. https://clintonhealthaccess.org/wp-content/uploads/2017/09/2017-ARV-Mark....
-
- Medicines Patent Pool. Press Release: Five Years On, 3.9 Million People in the Developing World Have Access to HIV Treatment Dolutegravir, Thanks to Access-Oriented Voluntary Licensing Agreements. https://medicinespatentpool.org/news-publications-post/five-years-on-3-9....
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

