Saving time maintaining reliability: a new method for quantification of Tetranychus urticae damage in Arabidopsis whole rosettes
- PMID: 32854637
- PMCID: PMC7450957
- DOI: 10.1186/s12870-020-02584-0
Saving time maintaining reliability: a new method for quantification of Tetranychus urticae damage in Arabidopsis whole rosettes
Abstract
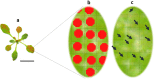
Background: The model species Tetranychus urticae produces important plant injury and economic losses in the field. The current accepted method for the quantification of the spider mite damage in Arabidopsis whole rosettes is time consuming and entails a bottleneck for large-scale studies such as mutant screening or quantitative genetic analyses. Here, we describe an improved version of the existing method by designing an automatic protocol. The accuracy, precision, reproducibility and concordance of the new enhanced approach are validated in two Arabidopsis accessions with opposite damage phenotypes. Results are compared to the currently available manual method.
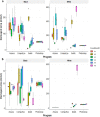
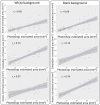
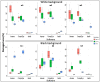
Results: Image acquisition experiments revealed that the automatic settings plus 10 values of brightness and the black background are the optimal conditions for a specific recognition of spider mite damage by software programs. Among the different tested methods, the Ilastik-Fiji tandem based on machine learning was the best procedure able to quantify the damage maintaining the differential range of damage between accessions. In addition, the Ilastik-Fiji tandem method showed the lowest variability within a set of conditions and the highest stability under different lighting or background surroundings. Bland-Altman concordance results pointed out a negative value for Ilastik-Fiji, which implies a minor estimation of the damage when compared to the manual standard method.
Conclusions: The novel approach using Ilastik and Fiji programs entails a great improvement for the quantification of the specific spider mite damage in Arabidopsis whole rosettes. The automation of the proposed method based on interactive machine learning eliminates the subjectivity and inter-rater-variability of the previous manual protocol. Besides, this method offers a robust tool for time saving and to avoid the damage overestimation observed with other methods.
Keywords: Arabidopsis thaliana; Assess; Chlorotic spots; CompuEye; Fiji; Ilastik; Machine learning; Photoshop; Plant damage quantification; Tetranychus urticae.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures







References
-
- Luedeling E, Steinmann KP, Zhang M, Brown PH, Grant J, Girvetz EH, et al. Climate change effects on walnut pests in California. Glob Chang Biol. 2011;17:228–238.
-
- Van Leeuwen T, Dermauw W. The molecular evolution of xenobiotic metabolism and resistance in Chelicerate mites. Annu Rev Entomol. 2016;61:475–498. - PubMed
-
- Park YL, Lee JH. Leaf cell and tissue damage of cucumber caused by two spotted spider mite (Acari: Tetranychidae) J Econ Entomol. 2002;95:952–957. - PubMed
-
- Farouk S, Osman MA. The effect of plant defence elicitors on common bean (Phaseolus vulgaris L.) growth and yield in absence or presence of spider mite (Tetranychus urticae Koch) infestation. J Stress Physiol Biochem. 2011;7:5–22.
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

