MMP-13 enzyme and pH responsive theranostic nanoplatform for osteoarthritis
- PMID: 32854712
- PMCID: PMC7450974
- DOI: 10.1186/s12951-020-00666-7
MMP-13 enzyme and pH responsive theranostic nanoplatform for osteoarthritis
Abstract
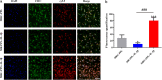
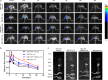
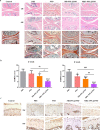
Stimulus-responsive therapy permits precise control of therapeutic effect only at lesion of interest, which determines it a promising method for diagnosis and imaging-guided precision therapy. The acid environment and overexpressed matrix metalloproteinases-13 (MMP-13) are typical markers in osteoarthritis (OA), which enables the development of stimulus-responsive drug delivery system with high specificity for OA. We herein demonstrate a nano-micelle based stimuli-responsive theranostic strategy with reporting and drug release controlled by acidic pH and MMP-13 for OA therapy. Such nanoplatform is incorporated with a motif specifically targeting on cartilage, a motif responsive to matrix metalloproteinases-13 to specifically report OA condition and biodynamics of nano-micelles, an anti-inflammatory drug (e.g., psoralidin (PSO)) from traditional Chinese medicine, and a biocompatible polymeric skeleton for sustainable drug release in response to the acidic OA condition. The high effectiveness of this targeted precision therapy is demonstrated comprehensively by both in vitro and vivo evidences.
Keywords: Cartilage targeting; MMP-13/pH sensitive; Osteoarthritis; Theranostics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Bender B, Perry M, Ramsey F, Boeselager G, Jackson-Thompson J. Prevalence and impact of chronic joint symptoms. J Am Med Assoc. 1998;279:1940–1941. doi: 10.1001/jama.279.24.1940. - DOI
-
- Brown TJ, Laurent UB, Fraser: Turnover of hyaluronan in synovial joints: elimination of labelled hyaluronan from the knee joint of the rabbit. Experimental Physiology 1991, 76. - PubMed
MeSH terms
Substances
Grants and funding
- 2018YFC1105900/National key research and development program of China
- 2018YFC1105900/National Key R&D Program of China
- 81960414/National Natural Science Fund of China
- GuikeAD17129012/the Guangxi Science and Technology Base and Talent Special Project
- GuikeZY18164004/the local Science and Technology Development Project leading by the central government
- 2019KY0134/the Basic ability enhancement project for young and middle-aged teachers of universities in Guangxi
- 2018KY0127/the Basic ability enhancement project for young and middle-aged teachers of universities in Guangxi
- SG2018005/nd the Innovative Project of Postgraduate Majored in Biomedical Engineering
- SG2018007/the Innovative Project of Postgraduate Majored in Biomedical Engineering
LinkOut - more resources
Full Text Sources
Medical

