The Regenerative Effect of Trans-spinal Magnetic Stimulation After Spinal Cord Injury: Mechanisms and Pathways Underlying the Effect
- PMID: 32856173
- PMCID: PMC7851265
- DOI: 10.1007/s13311-020-00915-5
The Regenerative Effect of Trans-spinal Magnetic Stimulation After Spinal Cord Injury: Mechanisms and Pathways Underlying the Effect
Abstract
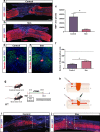
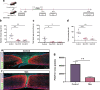
Spinal cord injury (SCI) leads to a loss of sensitive and motor functions. Currently, there is no therapeutic intervention offering a complete recovery. Here, we report that repetitive trans-spinal magnetic stimulation (rTSMS) can be a noninvasive SCI treatment that enhances tissue repair and functional recovery. Several techniques including immunohistochemical, behavioral, cells cultures, and proteomics have been performed. Moreover, different lesion paradigms, such as acute and chronic phase following SCI in wild-type and transgenic animals at different ages (juvenile, adult, and aged), have been used. We demonstrate that rTSMS modulates the lesion scar by decreasing fibrosis and inflammation and increases proliferation of spinal cord stem cells. Our results demonstrate also that rTSMS decreases demyelination, which contributes to axonal regrowth, neuronal survival, and locomotor recovery after SCI. This research provides evidence that rTSMS induces therapeutic effects in a preclinical rodent model and suggests possible translation to clinical application in humans.
Keywords: Rehabilitation; glial scar; magnetic stimulation and neuroregeneration; spinal cord injury.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures






References
-
- Wilson JR, Cadotte DW, Fehlings MG. Clinical predictors of neurological outcome, functional status, and survival after traumatic spinal cord injury: a systematic review. J Neurosurg Spine. 2012;17:11–26. - PubMed
-
- Siddiqui AM, Khazaei M, Fehlings MG. Translating mechanisms of neuroprotection, regeneration, and repair to treatment of spinal cord injury. Progress in Brain Research. 2015;218:15–54. - PubMed
-
- Goritz C, Dias DO, Tomilin N, et al. A Pericyte Origin of Spinal Cord Scar Tissue. Science. 2011;333:238–242. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

