Maladaptive Changes Associated With Cardiac Aging Are Sex-Specific and Graded by Frailty and Inflammation in C57BL/6 Mice
- PMID: 32857156
- PMCID: PMC7812442
- DOI: 10.1093/gerona/glaa212
Maladaptive Changes Associated With Cardiac Aging Are Sex-Specific and Graded by Frailty and Inflammation in C57BL/6 Mice
Abstract
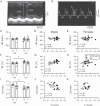
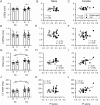
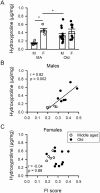
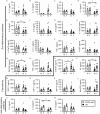
We investigated whether late-life changes in cardiac structure and function were related to high levels of frailty and inflammation in male and female mice. Frailty (frailty index), ventricular structure/function (echocardiography), and serum cytokines (multiplex immunoassay) were measured in 16- and 23-month-old mice. Left ventricular (LV) mass and septal wall thickness increased with age in both sexes. Ejection fraction increased with age in males (60.4 ± 1.4 vs 68.9 ± 1.8%; p < .05) but not females (58.8 ± 2.5 vs 62.6 ± 2.4%). E/A ratios declined with age in males (1.6 ± 0.1 vs 1.3 ± 0.1; p < .05) but not females (1.4 ± 0.1 vs 1.3 ± 0.1) and this was accompanied by increased ventricular collagen levels in males. These changes in ejection fraction (r = 0.52; p = .01), septal wall thickness (r = 0.59; p = .002), E/A ratios (r = -0.49; p = .04), and fibrosis (r = 0.82; p = .002) were closely graded by frailty scores in males. Only septal wall thickness and LV mass increased with frailty in females. Serum cytokines changed modestly with age in both sexes. Nonetheless, in males, E/A ratios, LV mass, LV posterior wall thickness, and septal wall thickness increased as serum cytokines increased (eg, IL-6, IL-3, IL-1α, IL-1β, tumor necrosis factor-α, eotaxin, and macrophage inflammatory protein-1α), while ejection fraction declined with increasing IL-3 and granulocyte-macrophage colony stimulating factor. Cardiac outcomes were not correlated with inflammatory cytokines in females. Thus, changes in cardiac structure and function in late life are closely graded by both frailty and markers of inflammation, but this occurs primarily in males. This suggests poor overall health and inflammation drive maladaptive changes in older male hearts, while older females may be resistant to these adverse effects of frailty.
Keywords: Chemokines; Echocardiography; Frailty index; Pro-inflammatory cytokines; Sex differences.
© The Author(s) 2020. Published by Oxford University Press on behalf of The Gerontological Society of America.
Figures
Similar articles
-
The impact of age and frailty on ventricular structure and function in C57BL/6J mice.J Physiol. 2017 Jun 15;595(12):3721-3742. doi: 10.1113/JP274134. Epub 2017 May 14. J Physiol. 2017. PMID: 28502095 Free PMC article.
-
Age- and sex-dependent differences in extracellular matrix metabolism associate with cardiac functional and structural changes.J Mol Cell Cardiol. 2020 Feb;139:62-74. doi: 10.1016/j.yjmcc.2020.01.005. Epub 2020 Jan 22. J Mol Cell Cardiol. 2020. PMID: 31978395 Free PMC article.
-
Aerobic Exercise Attenuates Frailty in Aging Male and Female C57Bl/6 Mice and Effects Systemic Cytokines Differentially by Sex.J Gerontol A Biol Sci Med Sci. 2022 Jan 7;77(1):41-46. doi: 10.1093/gerona/glab297. J Gerontol A Biol Sci Med Sci. 2022. PMID: 34610102 Free PMC article.
-
Sex-specific effects of frailty on cardiac structure and function: insights from preclinical models.Can J Physiol Pharmacol. 2024 Aug 1;102(8):476-486. doi: 10.1139/cjpp-2024-0009. Epub 2024 Mar 15. Can J Physiol Pharmacol. 2024. PMID: 38489788 Review.
-
Frailty and cytokines in preclinical models: Comparisons with humans.Mech Ageing Dev. 2022 Sep;206:111706. doi: 10.1016/j.mad.2022.111706. Epub 2022 Jul 12. Mech Ageing Dev. 2022. PMID: 35835224 Review.
Cited by
-
Correlation between frailty and cardiac structure and function in echocardiography in elderly patients with normal ejection fraction.Aging Clin Exp Res. 2023 Apr;35(4):775-784. doi: 10.1007/s40520-023-02363-5. Epub 2023 Mar 5. Aging Clin Exp Res. 2023. PMID: 36871112
-
The Impact of Melatonin Supplementation and NLRP3 Inflammasome Deletion on Age-Accompanied Cardiac Damage.Antioxidants (Basel). 2021 Aug 10;10(8):1269. doi: 10.3390/antiox10081269. Antioxidants (Basel). 2021. PMID: 34439517 Free PMC article.
-
Physical Interventions Restore Physical Frailty and the Expression of CXCL-10 and IL-1β Inflammatory Biomarkers in Old Individuals and Mice.Biomolecules. 2024 Jan 31;14(2):166. doi: 10.3390/biom14020166. Biomolecules. 2024. PMID: 38397403 Free PMC article.
-
Cardiac response to adrenergic stress differs by sex and across the lifespan.Geroscience. 2021 Aug;43(4):1799-1813. doi: 10.1007/s11357-021-00345-x. Epub 2021 Mar 2. Geroscience. 2021. PMID: 33651247 Free PMC article.
-
Therapeutic potential and mechanistic insights of astragaloside IV in the treatment of arrhythmia: a comprehensive review.Front Pharmacol. 2025 Apr 10;16:1528208. doi: 10.3389/fphar.2025.1528208. eCollection 2025. Front Pharmacol. 2025. PMID: 40276608 Free PMC article. Review.
References
-
- Ghimire A, Kane AE, Howlett SE. Sex differences in the physiology and pathology of the aging heart. In: Sawyer D, Vasan R, eds. Encyclopedia of Cardiovascular Research and Medicine. Amsterdam, The Netherlands: Elsevier; 2018:368–376.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

