Direct-from-sputum rapid phenotypic drug susceptibility test for mycobacteria
- PMID: 32857802
- PMCID: PMC7454970
- DOI: 10.1371/journal.pone.0238298
Direct-from-sputum rapid phenotypic drug susceptibility test for mycobacteria
Abstract
Background: The spread of multi-drug resistant tuberculosis (MDR-TB) is a leading global public-health challenge. Because not all biological mechanisms of resistance are known, culture-based (phenotypic) drug-susceptibility testing (DST) provides important information that influences clinical decision-making. Current phenotypic tests typically require pre-culture to ensure bacterial loads are at a testable level (taking 2-4 weeks) followed by 10-14 days to confirm growth or lack thereof.
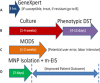
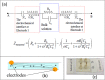
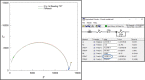
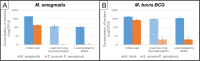
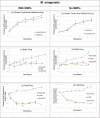
Methods and findings: We present a 2-step method to obtain DST results within 3 days of sample collection. The first involves selectively concentrating live mycobacterial cells present in relatively large volumes of sputum (~2-10mL) using commercially available magnetic-nanoparticles (MNPs) into smaller volumes, thereby bypassing the need for pre-culture. The second involves using microchannel Electrical Impedance Spectroscopy (m-EIS) to monitor multiple aliquots of small volumes (~10μL) of suspension containing mycobacterial cells, MNPs, and candidate-drugs to determine whether cells grow, die, or remain static under the conditions tested. m-EIS yields an estimate for the solution "bulk capacitance" (Cb), a parameter that is proportional to the number of live bacteria in suspension. We are thus able to detect cell death (bactericidal action of the drug) in addition to cell-growth. We demonstrate proof-of-principle using M. bovis BCG and M. smegmatis suspended in artificial sputum. Loads of ~ 2000-10,000 CFU of mycobacteria were extracted from ~5mL of artificial sputum during the decontamination process with efficiencies of 84% -100%. Subsequently, suspensions containing ~105 CFU/mL of mycobacteria with 10 mg/mL of MNPs were monitored in the presence of bacteriostatic and bactericidal drugs at concentrations below, at, and above known MIC (Minimum Inhibitory Concentration) values. m-EIS data (ΔCb) showed data consistent with growth, death or stasis as expected and/or recorded using plate counts. Electrical signals of death were visible as early as 3 hours, and growth was seen in < 3 days for all samples, allowing us to perform DST in < 3 days.
Conclusion: We demonstrated "proof of principle" that (a) live mycobacteria can be isolated from sputum using MNPs with high efficiency (almost all the bacteria that survive decontamination) and (b) that the efficacy of candidate drugs on the mycobacteria thus isolated (in suspensions containing MNPs) could be tested in real-time using m-EIS.
Conflict of interest statement
Shramik Sengupta owns equity interest in ImpeDx Diagnostics. ImpeDx has licensed technology from the University of Missouri. Shramik Sengupta, Sachidevi Puttaswamy and Roli Kargupta are co-inventors of this technology. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures



Similar articles
-
[Mycobacterial tests].Kekkaku. 2008 Jan;83(1):43-59. Kekkaku. 2008. PMID: 18283915 Japanese.
-
Rapid culture-based detection of living mycobacteria using microchannel electrical impedance spectroscopy (m-EIS).Biol Res. 2017 Jun 10;50(1):21. doi: 10.1186/s40659-017-0126-7. Biol Res. 2017. PMID: 28601089 Free PMC article.
-
[Improvement of routine works and quality control in mycobacterial laboratory].Kekkaku. 2007 Mar;82(3):217-27. Kekkaku. 2007. PMID: 17444126 Japanese.
-
Methods for determining the antimicrobial susceptibility of mycobacteria.Enferm Infecc Microbiol Clin. 2017 Oct;35(8):529-535. doi: 10.1016/j.eimc.2016.04.008. Epub 2016 May 25. Enferm Infecc Microbiol Clin. 2017. PMID: 27236235 Review. English, Spanish.
-
Mycobacterial Growth.Cold Spring Harb Perspect Med. 2015 May 8;5(10):a021097. doi: 10.1101/cshperspect.a021097. Cold Spring Harb Perspect Med. 2015. PMID: 25957314 Free PMC article. Review.
Cited by
-
Evolution of tuberculosis diagnostics: From molecular strategies to nanodiagnostics.Tuberculosis (Edinb). 2023 May;140:102340. doi: 10.1016/j.tube.2023.102340. Epub 2023 Apr 5. Tuberculosis (Edinb). 2023. PMID: 37031646 Free PMC article. Review.
-
QMAC-DST for Rapid Detection of Drug Resistance in Pulmonary Tuberculosis Patients: A Multicenter Pre-Post Comparative Study.J Clin Med. 2024 May 16;13(10):2941. doi: 10.3390/jcm13102941. J Clin Med. 2024. PMID: 38792481 Free PMC article.
References
-
- World_Health_Organization. Global Tuberculosis Report. 2018.
-
- Control UCfD, Prevention. Core curriculum on tuberculosis: what the clinician should know. Centers for Disease Control and Prevention Atlanta, GA; 2013.
-
- Organization WH. Global tuberculosis report 2015: World Health Organization; 2015.
-
- World_Health_Organization. Global MDR-TB situation; update 2017 (slides). 2017.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

