Phosphatidylserine controls calcium phosphate nucleation and growth on lipid monolayers: A physicochemical understanding of matrix vesicle-driven biomineralization
- PMID: 32858148
- PMCID: PMC8406674
- DOI: 10.1016/j.jsb.2020.107607
Phosphatidylserine controls calcium phosphate nucleation and growth on lipid monolayers: A physicochemical understanding of matrix vesicle-driven biomineralization
Abstract
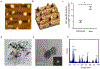
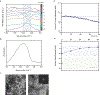
Bone biomineralization is an exquisite process by which a hierarchically organized mineral matrix is formed. Growing evidence has uncovered the involvement of one class of extracellular vesicles, named matrix vesicles (MVs), in the formation and delivery of the first mineral nuclei to direct collagen mineralization. MVs are released by mineralization-competent cells equipped with a specific biochemical machinery to initiate mineral formation. However, little is known about the mechanisms by which MVs can trigger this process. Here, we present a combination of in situ investigations and ex vivo analysis of MVs extracted from growing-femurs of chicken embryos to investigate the role played by phosphatidylserine (PS) in the formation of mineral nuclei. By using self-assembled Langmuir monolayers, we reconstructed the nucleation core - a PS-enriched motif thought to trigger mineral formation in the lumen of MVs. In situ infrared spectroscopy of Langmuir monolayers and ex situ analysis by transmission electron microscopy evidenced that mineralization was achieved in supersaturated solutions only when PS was present. PS nucleated amorphous calcium phosphate that converted into biomimetic apatite. By using monolayers containing lipids extracted from native MVs, mineral formation was also evidenced in a manner that resembles the artificial PS-enriched monolayers. PS-enrichment in lipid monolayers creates nanodomains for local increase of supersaturation, leading to the nucleation of ACP at the interface through a multistep process. We posited that PS-mediated nucleation could be a predominant mechanism to produce the very first mineral nuclei during MV-driven bone/cartilage biomineralization.
Keywords: Biomineralization; Calcium phosphate; Langmuir monolayers; Matrix vesicles; Phosphatidylserine.
Copyright © 2020 Elsevier Inc. All rights reserved.
Figures
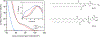

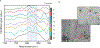
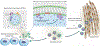
Similar articles
-
Enzymatic Approach in Calcium Phosphate Biomineralization: A Contribution to Reconcile the Physicochemical with the Physiological View.Int J Mol Sci. 2021 Nov 30;22(23):12957. doi: 10.3390/ijms222312957. Int J Mol Sci. 2021. PMID: 34884758 Free PMC article. Review.
-
Mineralization of annexin-5-containing lipid-calcium-phosphate complexes: modulation by varying lipid composition and incubation with cartilage collagens.J Biol Chem. 2008 Apr 11;283(15):9737-48. doi: 10.1074/jbc.M706523200. Epub 2008 Feb 4. J Biol Chem. 2008. PMID: 18250169 Free PMC article.
-
Quantitative atomic force microscopy provides new insight into matrix vesicle mineralization.Arch Biochem Biophys. 2019 May 30;667:14-21. doi: 10.1016/j.abb.2019.04.003. Epub 2019 Apr 16. Arch Biochem Biophys. 2019. PMID: 30998909 Free PMC article.
-
Differential effects of zinc and magnesium ions on mineralization activity of phosphatidylserine calcium phosphate complexes.J Inorg Biochem. 2009 Jul;103(7):948-62. doi: 10.1016/j.jinorgbio.2009.04.004. Epub 2009 Apr 19. J Inorg Biochem. 2009. PMID: 19477528
-
Matrix vesicles from chondrocytes and osteoblasts: Their biogenesis, properties, functions and biomimetic models.Biochim Biophys Acta Gen Subj. 2018 Mar;1862(3):532-546. doi: 10.1016/j.bbagen.2017.11.005. Epub 2017 Nov 3. Biochim Biophys Acta Gen Subj. 2018. PMID: 29108957 Free PMC article. Review.
Cited by
-
NPP1 and TNAP hydrolyze ATP synergistically during biomineralization.Purinergic Signal. 2023 Jun;19(2):353-366. doi: 10.1007/s11302-022-09882-2. Epub 2022 Jul 23. Purinergic Signal. 2023. PMID: 35870033 Free PMC article.
-
Matrix Vesicles: Role in Bone Mineralization and Potential Use as Therapeutics.Pharmaceuticals (Basel). 2021 Mar 24;14(4):289. doi: 10.3390/ph14040289. Pharmaceuticals (Basel). 2021. PMID: 33805145 Free PMC article. Review.
-
TNAP as a therapeutic target for cardiovascular calcification: a discussion of its pleiotropic functions in the body.Cardiovasc Res. 2022 Jan 7;118(1):84-96. doi: 10.1093/cvr/cvaa299. Cardiovasc Res. 2022. PMID: 33070177 Free PMC article. Review.
-
Evaluation of Current Studies to Elucidate Processes in Dental Follicle Cells Driving Osteogenic Differentiation.Biomedicines. 2023 Oct 13;11(10):2787. doi: 10.3390/biomedicines11102787. Biomedicines. 2023. PMID: 37893160 Free PMC article. Review.
-
Building a digital library on research into mineralizing vesicles: a systematic review-based approach.Biophys Rev. 2025 Feb 13;17(2):627-651. doi: 10.1007/s12551-025-01282-1. eCollection 2025 Apr. Biophys Rev. 2025. PMID: 40376417 Review.
References
-
- Anderson HC, 1995. Molecular Biology of Matrix Vesicles. Clin Orthop Relat Res 314, 266–80. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

