A genetic predictive model for precision treatment of diffuse large B-cell lymphoma with early progression
- PMID: 32864130
- PMCID: PMC7448459
- DOI: 10.1186/s40364-020-00214-3
A genetic predictive model for precision treatment of diffuse large B-cell lymphoma with early progression
Abstract
Background: Early progression after the first-line R-CHOP treatment leads to a very dismal outcome and necessitates alternative treatment for patients with diffuse large B-cell lymphoma (DLBCL). This study aimed to develop a genetic predictive model for early progression and evaluate its potential in advancing alternative treatment.
Methods: Thirty-two hotspot driver genes were examined in 145 DLBCL patients and 5 DLBCL cell lines using next-generation sequencing. The association of clinical features, cell-of-origin, double expression, positive p53 protein, and gene alterations with early progression was analyzed, and the genetic predictive model was developed based on the related independent variables and assessed by the area under receiver operating characteristic. The potential of novel treatment based on the modeling was investigated in in-vitro DLBCL cell lines and in vivo xenograft mouse models.
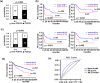
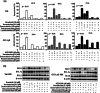
Results: The frequency of CD79B (42.86% vs 9.38%, p = 0.000) and PIM1 mutations (38.78% vs 17.71%, p = 0.005) showed a significant increase in patients with early progression. CD79B and PIM1 mutations were associated with complex genetic events, double expression, non-GCB subtype, advance stage and unfavorable prognosis. A powerful genetic predictive model (AUROC = 0.771, 95% CI: 0.689-0.853) incorporating lactate dehydrogenase levels (OR = 2.990, p = 0.018), CD79B mutations (OR = 5.970, p = 0.001), and PIM1 mutations (OR = 3.021, p = 0.026) was created and verified in the other cohort. This modeling for early progression outperformed the prediction accuracy of conventional International Prognostic Index, and new molecular subtypes of MCD and Cluster 5. CD79B and PIM1 mutations indicated a better response to inhibitors of BTK (ibrutinib) and pan-PIM kinase (AZD 1208) through repressing activated oncogenic signaling. Since the two inhibitors failed to decrease BCL2 level, BCL2 inhibitor (venetoclax) was added and demonstrated to enhance their apoptosis-inducing activity in mutant cells with double expression.
Conclusions: The genetic predictive model provides a robust tool to identify early progression and determine precision treatment. These findings warrant the development of optimal alternative treatment in clinical trials.
Keywords: CD79B; Diffuse large B-cell lymphoma; Early progression; PIM1.
© The Author(s) 2020.
Conflict of interest statement
Competing interestsAll of the authors declare no competing interests.
Figures




Similar articles
-
Deciphering the Prognostic Significance of MYD88 and CD79B Mutations in Diffuse Large B-Cell Lymphoma: Insights into Treatment Outcomes.Target Oncol. 2024 May;19(3):383-400. doi: 10.1007/s11523-024-01057-w. Epub 2024 Apr 21. Target Oncol. 2024. PMID: 38643457
-
The role of PIM1 in the ibrutinib-resistant ABC subtype of diffuse large B-cell lymphoma.Am J Cancer Res. 2016 Nov 1;6(11):2489-2501. eCollection 2016. Am J Cancer Res. 2016. PMID: 27904766 Free PMC article.
-
Molecular subtyping of CD5+ diffuse large B-cell lymphoma based on DNA-targeted sequencing and Lymph2Cx.Front Oncol. 2022 Aug 23;12:941347. doi: 10.3389/fonc.2022.941347. eCollection 2022. Front Oncol. 2022. PMID: 36081566 Free PMC article.
-
MYD88L265P and CD79B double mutations type (MCD type) of diffuse large B-cell lymphoma: mechanism, clinical characteristics, and targeted therapy.Ther Adv Hematol. 2022 Jan 31;13:20406207211072839. doi: 10.1177/20406207211072839. eCollection 2022. Ther Adv Hematol. 2022. PMID: 35126963 Free PMC article. Review.
-
Oncogenic Mutations of MYD88 and CD79B in Diffuse Large B-Cell Lymphoma and Implications for Clinical Practice.Cancers (Basel). 2020 Oct 10;12(10):2913. doi: 10.3390/cancers12102913. Cancers (Basel). 2020. PMID: 33050534 Free PMC article. Review.
Cited by
-
Tumor mutation burden estimated by a 69-gene-panel is associated with overall survival in patients with diffuse large B-cell lymphoma.Exp Hematol Oncol. 2021 Mar 15;10(1):20. doi: 10.1186/s40164-021-00215-4. Exp Hematol Oncol. 2021. PMID: 33722306 Free PMC article.
-
Machine-learning driven strategies for adapting immunotherapy in metastatic NSCLC.Nat Commun. 2025 Jul 24;16(1):6828. doi: 10.1038/s41467-025-61823-w. Nat Commun. 2025. PMID: 40707438 Free PMC article.
-
Oncogenic Mutations and Tumor Microenvironment Alterations of Older Patients With Diffuse Large B-Cell Lymphoma.Front Immunol. 2022 Mar 25;13:842439. doi: 10.3389/fimmu.2022.842439. eCollection 2022. Front Immunol. 2022. PMID: 35401516 Free PMC article.
-
A 6-tsRNA signature for early detection, treatment response monitoring, and prognosis prediction in diffuse large B cell lymphoma.Blood Cancer J. 2025 Apr 28;15(1):79. doi: 10.1038/s41408-025-01267-z. Blood Cancer J. 2025. PMID: 40295511 Free PMC article.
-
The Addition of Ferritin Enhanced the Prognostic Value of International Prognostic Index in Diffuse Large B-Cell Lymphoma.Front Oncol. 2022 Jan 19;11:823079. doi: 10.3389/fonc.2021.823079. eCollection 2021. Front Oncol. 2022. PMID: 35127536 Free PMC article.
References
-
- Coiffier B, et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the Groupe d'Etudes des Lymphomes de l'Adulte. Blood. 2010;116(12):2040–2045. doi: 10.1182/blood-2010-03-276246. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

