Rethinking the Nitrogenase Mechanism: Activating the Active Site
- PMID: 32864580
- PMCID: PMC7451245
- DOI: 10.1016/j.joule.2019.09.004
Rethinking the Nitrogenase Mechanism: Activating the Active Site
Abstract
Metalloenzymes called nitrogenases (N2ases) harness the reactivity of transition metals to reduce N2 to NH3. Specifically, N2ases feature a multimetallic active site, called a cofactor, which binds and reduces N2. The seven Fe centers and one additional metal center (Mo, V, or Fe) that make up the cofactor are all potential substrate binding sites. Unraveling the mechanism by which the cofactor binds N2 and reduces N2 to NH3 represents a multifaceted challenge because cofactor activation is required for N2 binding and functionalization to NH3. Despite decades of fascinating contributions, the nature of N2 binding to the active site and the structure of the activated cofactor remain unknown. Herein, we discuss the challenges associated with N2 reduction and how transition metal complexes facilitate N2 functionalization by coordinating N2. We also review the activation and/or reaction mechanisms reported for small molecule catalysts and the Haber-Bosch catalyst and discuss their potential relevance to biological N2 fixation. Finally, we survey what is known about the mechanism of N2ase and highlight recent X-ray crystallographic studies supporting Fe-S bond cleavage at the active site to generate reactive Fe centers as a potential, underexplored route for cofactor activation. We propose that structural rearrangements, beyond electron and proton transfers, are key in generating the catalytically active state(s) of the cofactor. Understanding the mechanism of activation will be key to understanding N2 binding and reduction.
Conflict of interest statement
Declaration of Interests The authors declare no competing interests.
Figures


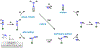
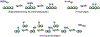

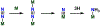
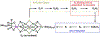

References
-
- Smil V (2001). Enriching the Earth : Fritz Haber, Carl Bosch, and the transformation of world food production (The MIT Press; ).
-
- Jennings JR (1991). Catalytic ammonia synthesis: fundamentals and practice (Springer Science+Business Media; ).
-
- Howard JB, and Rees DC (1996). Structural basis of biological nitrogen fixation. Chem. Rev 96, 2965–2982. - PubMed
-
- Burgess BK, and Lowe DJ (1996). Mechanism of molybdenum nitrogenase. Chem. Rev 96, 2983–3012. - PubMed
-
- Studt F, and Tuczek F (2005). Energetics and mechanism of a room-temperature catalytic process for ammonia synthesis (Schrock cycle): comparison with biological nitrogen fixation. Angew. Chem. Int. Ed 44, 5639–5642. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
