Hydrophobic interactions between the HA helix and S4-S5 linker modulate apparent Ca2+ sensitivity of SK2 channels
- PMID: 32865319
- PMCID: PMC7736289
- DOI: 10.1111/apha.13552
Hydrophobic interactions between the HA helix and S4-S5 linker modulate apparent Ca2+ sensitivity of SK2 channels
Abstract
Aim: Small-conductance Ca2+ -activated potassium (SK) channels are activated exclusively by increases in intracellular Ca2+ that binds to calmodulin constitutively associated with the channel. Wild-type SK2 channels are activated by Ca2+ with an EC50 value of ~0.3 μmol/L. Here, we investigate hydrophobic interactions between the HA helix and the S4-S5 linker as a major determinant of channel apparent Ca2+ sensitivity.
Methods: Site-directed mutagenesis, electrophysiological recordings and molecular dynamic (MD) simulations were utilized.
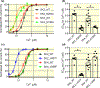
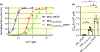
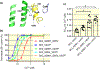
Results: Mutations that decrease hydrophobicity at the HA-S4-S5 interface lead to Ca2+ hyposensitivity of SK2 channels. Mutations that increase hydrophobicity result in hypersensitivity to Ca2+ . The Ca2+ hypersensitivity of the V407F mutant relies on the interaction of the cognate phenylalanine with the S4-S5 linker in the SK2 channel. Replacing the S4-S5 linker of the SK2 channel with the S4-S5 linker of the SK4 channel results in loss of the hypersensitivity caused by V407F. This difference between the S4-S5 linkers of SK2 and SK4 channels can be partially attributed to I295 equivalent to a valine in the SK4 channel. A N293A mutation in the S4-S5 linker also increases hydrophobicity at the HA-S4-S5 interface and elevates the channel apparent Ca2+ sensitivity. The double N293A/V407F mutations generate a highly Ca2+ sensitive channel, with an EC50 of 0.02 μmol/L. The MD simulations of this double-mutant channel revealed a larger channel cytoplasmic gate.
Conclusion: The electrophysiological data and MD simulations collectively suggest a crucial role of the interactions between the HA helix and S4-S5 linker in the apparent Ca2+ sensitivity of SK2 channels.
Keywords: HA helix; S4-S5 linker; SK2 channels.
© 2020 Scandinavian Physiological Society. Published by John Wiley & Sons Ltd.
Conflict of interest statement
Figures




Comment in
-
Deciphering the molecular mechanism of SK2 channel activation by intracellular calcium to develop new therapeutic agents.Acta Physiol (Oxf). 2021 Jan;231(1):e13574. doi: 10.1111/apha.13574. Epub 2020 Nov 30. Acta Physiol (Oxf). 2021. PMID: 33119956 No abstract available.
Similar articles
-
Channelopathy of small- and intermediate-conductance Ca2+-activated K+ channels.Acta Pharmacol Sin. 2023 Feb;44(2):259-267. doi: 10.1038/s41401-022-00935-1. Epub 2022 Jun 17. Acta Pharmacol Sin. 2023. PMID: 35715699 Free PMC article. Review.
-
The concerted contribution of the S4-S5 linker and the S6 segment to the modulation of a Kv channel by 1-alkanols.Mol Pharmacol. 2006 Nov;70(5):1542-54. doi: 10.1124/mol.106.026187. Epub 2006 Aug 3. Mol Pharmacol. 2006. PMID: 16887933
-
Role of amino-terminal half of the S4-S5 linker in type 1 ryanodine receptor (RyR1) channel gating.J Biol Chem. 2011 Oct 14;286(41):35571-35577. doi: 10.1074/jbc.M111.255240. Epub 2011 Aug 23. J Biol Chem. 2011. PMID: 21862589 Free PMC article.
-
Allosteric inhibitors targeting the calmodulin-PIP2 interface of SK4 K+ channels for atrial fibrillation treatment.Proc Natl Acad Sci U S A. 2022 Aug 23;119(34):e2202926119. doi: 10.1073/pnas.2202926119. Epub 2022 Aug 15. Proc Natl Acad Sci U S A. 2022. PMID: 35969786 Free PMC article.
-
The S4---S5 linker - gearbox of TRP channel gating.Cell Calcium. 2017 Nov;67:156-165. doi: 10.1016/j.ceca.2017.04.002. Epub 2017 Apr 4. Cell Calcium. 2017. PMID: 28416203 Review.
Cited by
-
Channelopathy of small- and intermediate-conductance Ca2+-activated K+ channels.Acta Pharmacol Sin. 2023 Feb;44(2):259-267. doi: 10.1038/s41401-022-00935-1. Epub 2022 Jun 17. Acta Pharmacol Sin. 2023. PMID: 35715699 Free PMC article. Review.
-
KCa2 and KCa3.1 Channels in the Airways: A New Therapeutic Target.Biomedicines. 2023 Jun 21;11(7):1780. doi: 10.3390/biomedicines11071780. Biomedicines. 2023. PMID: 37509419 Free PMC article. Review.
-
Structure-Activity Relationship Study of Subtype-Selective Positive Modulators of KCa2 Channels.J Med Chem. 2022 Jan 13;65(1):303-322. doi: 10.1021/acs.jmedchem.1c01473. Epub 2021 Dec 28. J Med Chem. 2022. PMID: 34962403 Free PMC article.
-
Subtype-Selective Positive Modulation of KCa2.3 Channels Increases Cilia Length.ACS Chem Biol. 2022 Aug 19;17(8):2344-2354. doi: 10.1021/acschembio.2c00469. Epub 2022 Aug 10. ACS Chem Biol. 2022. PMID: 35947779 Free PMC article.
-
Subtype-selective positive modulation of KCa 2 channels depends on the HA/HB helices.Br J Pharmacol. 2022 Feb;179(3):460-472. doi: 10.1111/bph.15676. Epub 2021 Oct 1. Br J Pharmacol. 2022. PMID: 34458981 Free PMC article.
References
-
- Adelman JP, Maylie J, Sah P. Small-conductance Ca2+-activated K+ channels: Form and function. Annu Rev Physiol. 2012;74:245–269. - PubMed
-
- Bildl W, Strassmaier T, Thurm H, et al. Protein kinase CK2 Is coassembled with small conductance Ca2+-activated K+ channels and regulates channel gating. Neuron. 2004;43(6):847–858. - PubMed
-
- Pedarzani P, Mosbacher J, Rivard A, et al. Control of Electrical Activity in Central Neurons by Modulating the Gating of Small Conductance Ca2+-activated K+ Channels. J Biol Chem. 2001;276(13):9762–9769. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

