Differential response of human T-lymphocytes to arsenic and uranium
- PMID: 32866568
- PMCID: PMC7590629
- DOI: 10.1016/j.toxlet.2020.08.013
Differential response of human T-lymphocytes to arsenic and uranium
Abstract
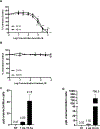
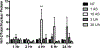
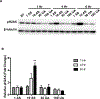
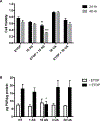
Elevated levels of arsenic and uranium have been detected in water sources near abandoned uranium mines in the Southwest. Evidence suggests uranium exposure increases the likelihood of immune dysfunction and this study investigates the impact of arsenic and uranium on human immune cell lines. Concentration-dependent cytotoxicity occurred following exposure to arsenite, whereas cells remained viable after 48 -h treatment with up to 100 μM uranyl acetate despite uptake of uranium into cells. Arsenite stimulated an oxidative stress response as detected by Nrf-2 nuclear accumulation and induction of HMOX-1 and NQO1, which was not detected with up to 30 μM uranyl acetate. Cellular oxidative stress can promote DNA damage and arsenite, but not uranium, stimulated DNA damage as measured by pH2AX. Arsenic enhanced the cytotoxic response to etoposide suggesting an inhibition of DNA repair, unlike uranium. Similarly, uranium did not inhibit PARP-1 activity. Because uranium reportedly stimulates oxidative stress, DNA damage and cytotoxicity in adherent epithelial cells, the current study suggests distinct cell type differences in response to uranium that may relate to generation of oxidative stress and associated downstream consequences. Delineating the actions of uranium across different cell targets will be important for understanding the potential health effects of uranium exposures.
Keywords: Arsenic; DNA damage; Immunotoxicity; Oxidative stress; T-lymphocyte; Uranium.
Copyright © 2020 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors report no declarations of interest.
Figures




References
-
- Agency for Toxic Substances & Disease Registry, Toxicological Profile: Arsenic, (2007). http://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=22&tid=3#bookmark07 (accessed July 21, 2011). - PubMed
-
- Agency for Toxic Substances and Disease Registry (ATSDR), Toxicological profile for Uranium., (2013). https://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=440&tid=77 (accessed December 5, 2018). - PubMed
-
- Agency for Toxic Substances and Disease Registry (ATSDR), Addendum to the Toxicological Profile for Arsenic, (2016). https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=2&cad=rja&... (accessed March 13, 2019).
-
- Blake JM, De Vore CL, Avasarala S, Ali A-M, Roldan C, Bowers F, Spilde MN, Artyushkova K, Kirk MF, Peterson E, Rodriguez-Freire L, Cerrato JM, Uranium mobility and accumulation along the Rio Paguate, Jackpile Mine in Laguna Pueblo, NM, Environmental Science: Processes & Impacts. 19 (2017) 605–621. 10.1039/C6EM00612D. - DOI - PubMed
-
- Eggers M, Doyle J, Lefthand M, Young S, Moore-Nall A, Kindness L, Other Medicine R, Ford T, Dietrich E, Parker A, Hoover J, Camper A, Community Engaged Cumulative Risk Assessment of Exposure to Inorganic Well Water Contaminants, Crow Reservation, Montana, International Journal of Environmental Research and Public Health. 15 (2018) 76 10.3390/ijerph15010076. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

