Pharmacologic and surgical therapies for patients with Meniere's disease: A systematic review and network meta-analysis
- PMID: 32870918
- PMCID: PMC7462264
- DOI: 10.1371/journal.pone.0237523
Pharmacologic and surgical therapies for patients with Meniere's disease: A systematic review and network meta-analysis
Abstract
Background: Meniere's disease (MD) is a chronic condition of the inner ear consisting of symptoms that include vertigo attacks, fluctuating sensorineural hearing loss, tinnitus and aural fullness. Despite availability of various interventions, there is uncertainty surrounding their relative efficacy, thus making it difficult to select the appropriate treatments for MD. The objective of this systematic review was to assess the relative effects of the available pharmacologic and surgical interventions in patients with MD with regard to vertigo and other key patient outcomes based on data from randomized clinical trials (RCTs).
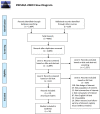
Methods: Our published protocol registered with PROSPERO (CRD42019119129) provides details on eligibility criteria and methods. We searched various databases including MEDLINE, Embase and the Cochrane Library from inception to December 10th, 2018. Screening at citation and full-text levels and risk of bias assessment were performed by two independent reviewers in duplicate, with discrepancies resolved by consensus or third-party adjudication. Bayesian network meta-analyses (NMA) were performed for hearing change and vertigo control outcomes, along with pairwise meta-analyses for these and additional outcomes.
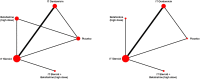
Results: We identified 2,889 unique citations, that yielded 23 relevant publications describing 18 unique RCTs (n = 1,231 patients). Overall, risk-of bias appraisal suggested the evidence base to be at unclear or high risk of bias. Amongst pharmacologics, we constructed treatment networks of five intervention groups that included placebo, intratympanic (IT) gentamicin, oral high-dose betahistine, IT steroid and IT steroid plus high-dose betahistine for NMAs of hearing change (improvement or deterioration) and complete vertigo control. IT steroid plus high-dose betahistine was associated with the largest difference in hearing improvement compared to placebo, followed by high-dose betahistine and IT steroid (though 95% credible intervals failed to rule out the possibility of no difference), while IT gentamicin was worse than IT steroid. The NMA of complete vertigo control suggested IT gentamicin was associated with the highest probability of achieving better complete vertigo control compared to placebo, followed by IT steroid plus high-dose betahistine. Only two studies related to surgical interventions were found, and data suggested no statistically significant difference in hearing changes between endolymphatic duct blockage (EDB) versus endolymphatic sac decompression (ESD), and ESD with or without steroid injection. One trial reported that 96.5% of patients in EDB group compared to 37.5% of the patients in ESD group achieved complete vertigo control 24 months after surgery (p = 0.002).
Conclusion: To achieve both hearing preservation and vertigo control, the best treatment option among the pharmacologic interventions compared may be IT steroid plus high-dose betahistine, considering that IT gentamicin may have good performance to control vertigo but may be detrimental to hearing preservation with high cumulative dosage and short interval between injections. However, IT steroid plus high-dose betahistine has not been compared in head-to-head trials against other interventions except for IT steroid alone in one trial, thus future trials that compare it with other interventions will help establish comparative effectiveness with direct evidence.
Conflict of interest statement
We have read the journal's policy and the authors of this manuscript have the following competing interests: BH has received consultancy fees from Eversana (previously Cornerstone Research Group) for methodologic advice related to systematic reviews and meta-analysis. This does not alter our adherence to PLOS ONE policies on sharing data and materials. The remaining authors declare that they have no competing interests.
Figures




References
-
- Canadian Hearing Society. Facts and Figures: Prevalence of hearing loss. [Internet]. https://www.chs.ca/facts-and-figures. 2017.
-
- Mathers C, Stein C, Ma Fat D, Rao C, Inoue M, Tomijima N, et al. Global burden of disease in the year 2000. [Internet]. http://www.who.int/healthinfo/paper50.pdf. 2002.
-
- Harkonen K, Kivekas I, Rautiainen M, Kotti V, Sivonen V, Vasama JP. Single-Sided Deafness: The Effect of Cochlear Implantation on Quality of Life, Quality of Hearing, and Working Performance. ORLJ Otorhinolaryngol Relat Spec. 2015;77:339–45. - PubMed
-
- Naude A. Clinical and audiological features of Meniere’s disease: Insight into the diagnostic process [Internet]. 2006. https://repository.up.ac.za/handle/2263/27851
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

