Investigation of glucose-6-phosphate dehydrogenase (G6PD) deficiency prevalence in a Plasmodium vivax-endemic area in the Republic of Korea (ROK)
- PMID: 32873296
- PMCID: PMC7465311
- DOI: 10.1186/s12936-020-03393-4
Investigation of glucose-6-phosphate dehydrogenase (G6PD) deficiency prevalence in a Plasmodium vivax-endemic area in the Republic of Korea (ROK)
Abstract
Background: Glucose-6-phosphate dehydrogenase (G6PD) deficiency is the most prevalent inborn disorder. This X-chromosome-linked recessive disease affects more than 400 million people globally, and is associated with haemolytic anaemia after medication with the anti-latent malaria drug, primaquine. To prevent malaria, the Republic of Korea (ROK) Army administers malaria chemoprophylaxis. Due to the previously low G6PD deficiency prevalence in the ROK, prior to primaquine administration, testing for G6PD deficiency was not mandatory. In this study, to evaluate the risk from malaria chemoprophylaxis in the ROK, G6PD deficiency prevalence was investigated.
Methods: Blood specimens from 1632 soldiers entering training camp for the 3rd Infantry of the ROK Army were collected. CareStart™ Biosensor for G6PD and haemoglobin (Hb) was used to detect G6PD levels. G6PD variants using the DiaPlexC G6PD Genotyping kit (Asian type) and full-length sequencing were examined.
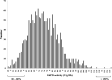
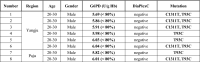
Results: Of 1632 blood specimens tested, none was observed to be G6PD deficient. The median value of all tested samples was 7.582 U/g Hb. An investigation of 170 G6PD DNA variants was analysed and categorized as partially low normal [n = 131, 30-80% (2.27-6.05 U/g Hb) of the median value], high [n = 3, > 150% (> 11.373 U/g Hb) of the median value], or normal [n = 36, 80-150% (6.05-11.373 U/g Hb) of the median value], and none was amplified by the DiaPlexC kit. Five silent mutations (C→T) in 131 partially low normal specimens were found at the 1311th nucleotide position by sequence analysis. Another 8 silent mutations (T93C) were also detected in 131 partially low normal specimens. Thus, it is inferred that these silent mutations could be related to G6PD activity.
Conclusions: This G6PD deficiency prevalence study, conducted among participants from the 3rd Infantry of the ROK Army, provided crucial evidence for the safety of malaria chemoprophylaxis. This study showed that the prevalence of G6PD deficiency among 1632 young soldiers was wholly absent. Although G6PD phenotypic mutations were not detected, many silent mutations (C1311T and T93C) were observed. Thus, it is inferred that malaria chemoprophylaxis is relatively safe against G6PD deficiency-mediated haemolytic anaemia. However, given the number of individuals whose G6PD were at the partially low normal range and the frequent detection of G6PD deficiency-related mutations, consistent monitoring of G6PD deficiency is needed.
Keywords: Glucose-6-phosphate dehydrogenase deficiency; Prevalence; Primaquine; Single nucleotide polymorphism.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
First evaluation of glucose-6-phosphate dehydrogenase (G6PD) deficiency in vivax malaria endemic regions in the Republic of Korea.PLoS One. 2014 May 22;9(5):e97390. doi: 10.1371/journal.pone.0097390. eCollection 2014. PLoS One. 2014. PMID: 24853873 Free PMC article.
-
Tolerability and safety of weekly primaquine against relapse of Plasmodium vivax in Cambodians with glucose-6-phosphate dehydrogenase deficiency.BMC Med. 2015 Aug 25;13:203. doi: 10.1186/s12916-015-0441-1. BMC Med. 2015. PMID: 26303162 Free PMC article.
-
G6PD deficiency in malaria endemic areas of Nepal.Malar J. 2020 Aug 12;19(1):287. doi: 10.1186/s12936-020-03359-6. Malar J. 2020. PMID: 32787970 Free PMC article.
-
Primaquine alternative dosing schedules for preventing malaria relapse in people with Plasmodium vivax.Cochrane Database Syst Rev. 2020 Aug 19;8:CD012656. doi: 10.1002/14651858.CD012656.pub3. Cochrane Database Syst Rev. 2020. PMID: 32816320
-
G6PD deficiency: global distribution, genetic variants and primaquine therapy.Adv Parasitol. 2013;81:133-201. doi: 10.1016/B978-0-12-407826-0.00004-7. Adv Parasitol. 2013. PMID: 23384623 Review.
Cited by
-
Cost-Benefit Analysis of Tafenoquine for Radical Cure of Plasmodium vivax Malaria in Korea.J Korean Med Sci. 2022 Jul 11;37(27):e212. doi: 10.3346/jkms.2022.37.e212. J Korean Med Sci. 2022. PMID: 35818703 Free PMC article.
-
Suboptimal Doses of Antimalarials Relative to Increasing Body Weight and the Risk of Plasmodium vivax Recurrence in the Republic of Korea Armed Forces, 2012-2021.J Korean Med Sci. 2024 Dec 23;39(49):e314. doi: 10.3346/jkms.2024.39.e314. J Korean Med Sci. 2024. PMID: 39716862 Free PMC article.
-
Performance of a Commercial Multiplex Allele-Specific Polymerase Chain Reaction Kit to Genotype African-Type Glucose-6-Phosphate Dehydrogenase Deficiency.Am J Trop Med Hyg. 2022 Dec 19;108(2):449-455. doi: 10.4269/ajtmh.21-1081. Print 2023 Feb 1. Am J Trop Med Hyg. 2022. PMID: 36535256 Free PMC article.
-
Single nucleotide polymorphism analysis of pvmdr-1 in Plasmodium vivax isolated from military personnel of Republic of Korea in 2016 and 2017.Malar J. 2022 Jun 28;21(1):205. doi: 10.1186/s12936-022-04214-6. Malar J. 2022. PMID: 35765071 Free PMC article.
-
Utilization of Glucose-6-Phosphate Dehydrogenase Test and the Prevalence of Enzyme Deficiency in Korea.J Clin Med. 2023 Apr 28;12(9):3179. doi: 10.3390/jcm12093179. J Clin Med. 2023. PMID: 37176619 Free PMC article.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

