The factor VIII treatment history of non-severe hemophilia A
- PMID: 32877570
- PMCID: PMC7756346
- DOI: 10.1111/jth.15076
The factor VIII treatment history of non-severe hemophilia A
Abstract
Background: In patients with non-severe hemophilia A, we lack detailed knowledge on the timing of treatment with factor VIII (FVIII) concentrates. This knowledge could provide information about the expected treatment timing in patients with severe hemophilia A treated with non-replacement therapies.
Objective: To assess the FVIII treatment history in patients with non-severe hemophilia A.
Methods: Patients with non-severe hemophilia (baseline FVIII activity [FVIII:C] 2-40 IU/dL) were included from the INSIGHT study. The primary outcome was median age at first FVIII exposure (ED1). In a subgroup of patients for whom more detailed information was available, we analyzed the secondary outcomes: median age at first 20 EDs, annualized bleeding rate for all bleeds (ABR), joint bleeds (AJBR), and major spontaneous bleeds (ASmBR).
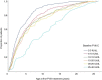
Results: In the total cohort (n = 1013), median baseline FVIII activity was 8 IU/dL (interquartile range [IQR] 4-15) and the median age at ED1 was 3.7 years (IQR 1.4-7.7). Median age at ED1 rose from 2.5 years (IQR 1.2-5.7) in patients with FVIII:C 2-5 IU/dL to 9.7 years (IQR 4.8-16.0) in patients with FVIII:C 25-40 IU/dL. In the subgroup (n = 104), median age at ED1, ED5, ED10, and ED20 was 4.0 years (IQR 1.4-7.6), 5.6 years (IQR 2.9-9.3), 7.5 years (IQR 4.4-11.3), and 10.2 years (IQR 6.5-14.2), respectively. Median ABR, AJBR, and ASmBR were 1.1 (IQR 0.5-2.6), 0.3 (IQR 0.1-0.7), and 0 (IQR 0-0), respectively.
Conclusion: This study demonstrates that in non-severe hemophilia A, the age at first FVIII exposure increases with baseline FVIII:C and that major spontaneous bleeds rarely occur.
Keywords: factor VIII; hemophilia A; hemorrhage; joint bleed; treatment.
© 2020 The Authors. Journal of Thrombosis and Haemostasis published by Wiley Periodicals LLC on behalf of International Society on Thrombosis and Haemostasis.
Conflict of interest statement
FK, AA, CE, EB, KP, CH, HP, JB, SG have nothing to disclose. CM has received research grant awards from Biotest and CSL Behring. He and/or his institution has received advisory of lecturing honoraria from Bayer, Biotest, CSL Behring, Novo Nordisk, Roche. GC received fees to act as a speaker at or to participate in Advisory Board meetings from Ablynx, Bayer, CSL Behring, Kedrion, Novo Nordisk, Shire/Takeda, Sobi, Roche, Uniqure, and Werfen. GC received unrestricted research grants from CSL Bhering, Pfizer, and Sobi. K. Fischer has received speaker's fees from Bayer, Baxter/Shire, Sobi/Biogen, CSL Behring, Octapharma, Pfizer, NovoNordisk, and has performed consultancy for Bayer, Baxter, Biogen, CSL‐Behring,Freeline, Novo Nordisk, Pfizer, Roche, and Sobi. She and/or her institution has received research support from Bayer, Pfizer, Baxter/Shire, and Novo Nordisk. MK received unrestricted research grants from ZonMW, Innovatiefonds, Bayer, Boehringer‐Ingelheim, Pfizer, Daiichi‐Sankyo for research outside of this work. MM received fees to act as an advisor/consultant/speaker for Bayer, CSL Behring, Novo Nordisk, Pfizer, Roche, Sobi, Bioverativ, Shire/Takeda, Octapharma, Kedrion, Grifols, Biotest, and Catalyst. CS has received consultancy or speaker fees by Bayer, CSL Behring, Shire/Takeda, Novo Nordisk, Sobi, Novartis, Pfizer, Amgen, and Roche. The institution of K. Fijnvandraat has received unrestricted research grants from CSL Behring, Novo Nordisk, and received consultancy fees from Grifols, Takeda, and Novo Nordisk. DPH has received research grant awards from Bayer, Octapharma, and Takeda. He and/or his institution have received advisory or lecturing honoraria from Bayer, Biomarin, Biotest, Grifols, Octapharma, Pfizer, Roche, Sanofi, Sobi, Takeda, UniQure.
Figures
References
-
- Fijnvandraat K, Cnossen MH, Leebeek FW, Peters M. Diagnosis and management of haemophilia. BMJ (Clinical research ed). 2012;344:e2707. - PubMed
-
- White GC 2nd, Rosendaal F, Aledort LM, Lusher JM, Rothschild C, Ingerslev J. Definitions in hemophilia. Recommendation of the scientific subcommittee on factor VIII and factor IX of the scientific and standardization committee of the International Society on Thrombosis and Haemostasis. Thromb Haemost. 2001;85(03):560. - PubMed
-
- Srivastava A, Brewer AK, Mauser‐Bunschoten EP, et al. Guidelines for the management of hemophilia. Haemophilia. 2013;19(1):e1‐e47. - PubMed
-
- Batty P, Austin SK, Khair K, et al. Treatment burden, haemostatic strategies and real world inhibitor screening practice in non‐severe haemophilia A. Br J Haematol. 2017;176(5):796‐804. - PubMed
-
- Donadel‐Claeyssens S. Current co‐ordinated activities of the PEDNET (European Paediatric Network for Haemophilia Management). Haemophilia. 2006;12(2):124‐127. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical