A Comparison of Intramuscular and Subcutaneous Administration of LigA Subunit Vaccine Adjuvanted with Neutral Liposomal Formulation Containing Monophosphoryl Lipid A and QS21
- PMID: 32882903
- PMCID: PMC7565420
- DOI: 10.3390/vaccines8030494
A Comparison of Intramuscular and Subcutaneous Administration of LigA Subunit Vaccine Adjuvanted with Neutral Liposomal Formulation Containing Monophosphoryl Lipid A and QS21
Abstract
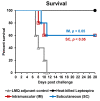
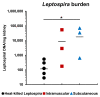
Leptospirosis vaccines with higher potency and reduced adverse effects are needed for human use. The carboxyl terminal domain of leptospiral immunoglobulin like protein A (LigAc) is currently the most promising candidate antigen for leptospirosis subunit vaccine. However, LigAc-based vaccines were unable to confer sterilizing immunity against Leptospira infection in animal models. Several factors including antigen properties, adjuvant, delivery system, and administration route need optimization to maximize vaccine efficacy. Our previous report demonstrated protective effects of the recombinant LigAc (rLigAc) formulated with liposome-based adjuvant, called LMQ (neutral liposome combined with monophosphoryl lipid A and Quillaja saponaria fraction 21) in hamsters. This study aimed to evaluate the impact of two commonly used administration routes, intramuscular (IM) and subcutaneous (SC), on immunogenicity and protective efficacy of rLigAc-LMQ administrated three times at 2-week interval. Two IM vaccinations triggered significantly higher levels of total anti-rLigAc IgG than two SC injections. However, comparable IgG titers and IgG2/IgG1 ratio was observed for both routes after the third immunization. The route of vaccine administration did not influence the survival rate (60%) and renal colonization against lethal Leptospira challenge. Importantly, the kidneys of IM group showed no pathological lesions while the SC group showed mild damage. In conclusion, IM vaccination with rLigAc-LMQ not only elicited faster antibody production but also protected from kidney damage following leptospiral infection better than SC immunization. However, both tested routes did not influence protective efficacy in terms of survival rate and the level of renal colonization.
Keywords: LMQ adjuvant; Leptospira; LigA subunit vaccine; intramuscular; leptospirosis; subcutaneous; vaccination route.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Grants and funding
- Southeast Asia-Europe Join Funding Scheme for Research and Innovation/Thailand Center of Excellence for Life Sciences
- Establishing a collaboration between the Chulalongkorn Vaccine Research Center (Chula VRC), Thailand and the Vaccine Formulation Laboratory (VFL), Switzerland, Barnier-Quer C. and Patarakul K./Swiss State Secretariat for Education, Research and Innovation (SERI) the Seed Money Project (ETH Zurich)
- Post-doctoral fellowship for Teerasit Techawiwattanaboon/Second Century Fund (C2F), Chulalongkorn University
LinkOut - more resources
Full Text Sources

