MicroRNA-132 attenuated cardiac fibrosis in myocardial infarction-induced heart failure rats
- PMID: 32885809
- PMCID: PMC7494995
- DOI: 10.1042/BSR20201696
MicroRNA-132 attenuated cardiac fibrosis in myocardial infarction-induced heart failure rats
Abstract
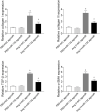
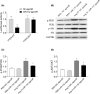
The aim of the present study was to determine the effect of microRNA (miR)-132 on cardiac fibrosis in myocardial infarction (MI)-induced heart failure and angiotensin (Ang) II-treated cardiac fibroblasts (CFs). Experiments were carried out in Sprague-Dawley rat treatment with ligation of left coronary artery to induce heart failure, and in CFs administration of Ang II to induce fibrosis. The level of miR-132 was increased in the heart of rats with MI-induced heart failure and the Ang II-treated CFs. In MI rats, left ventricle (LV) ejection fraction, fractional shortening, the maximum of the first differentiation of LV pressure (LV +dp/dtmax) and decline (LV -dp/dtmax) and LV systolic pressure (LVSP) were reduced, and LV end-systolic diameter (LVESD), LV end-diastolic diameter (LVEDD), LV volumes in systole (LVVS) and LV volumes in diastole (LVVD) were increased, which were reversed by miR-132 agomiR but deteriorated by miR-132 antagomiR. The expression levels of collagen I, collagen III, transforming growth factor-β (TGF-β), and α-smooth muscle actin (α-SMA) were increased in the heart of rat with MI-induced heart failure and CFs administration of Ang II. These increases were inhibited by miR-132 agomiR but enhanced by miR-132 antagomiR treatment. MiR-132 inhibited PTEN expression, and attenuated PI3K/Akt signal pathway in CFs. These results indicated that the up-regulation of miR-132 improved the cardiac dysfunction, attenuated cardiac fibrosis in heart failure via inhibiting PTEN expression, and attenuating PI3K/Akt signal pathway. Up-regulation of miR-132 may be a strategy for the treatment of heart failure and cardiac fibrosis.
Keywords: cardiac dysfunction; cardiac fibroblasts; fibrosis; heart failure; microRNA -132.
© 2020 The Author(s).
Conflict of interest statement
The authors declare that there are no competing interests associated with the manuscript.
Figures




Similar articles
-
Inhibition of microRNA-146a attenuated heart failure in myocardial infarction rats.Biosci Rep. 2019 Dec 20;39(12):BSR20191732. doi: 10.1042/BSR20191732. Biosci Rep. 2019. PMID: 31763669 Free PMC article.
-
Apelin-13 alleviated cardiac fibrosis via inhibiting the PI3K/Akt pathway to attenuate oxidative stress in rats with myocardial infarction-induced heart failure.Biosci Rep. 2020 Apr 30;40(4):BSR20200040. doi: 10.1042/BSR20200040. Biosci Rep. 2020. PMID: 32207519 Free PMC article.
-
Alarin alleviated cardiac fibrosis via attenuating oxidative stress in heart failure rats.Amino Acids. 2021 Jul;53(7):1079-1089. doi: 10.1007/s00726-021-03005-8. Epub 2021 Jun 5. Amino Acids. 2021. PMID: 34089389 Free PMC article.
-
Clinical aspects of left ventricular diastolic function assessed by Doppler echocardiography following acute myocardial infarction.Dan Med Bull. 2001 Nov;48(4):199-210. Dan Med Bull. 2001. PMID: 11767125 Review.
-
Inhibitory effect of microRNA-21 on pathways and mechanisms involved in cardiac fibrosis development.Ther Adv Cardiovasc Dis. 2024 Jan-Dec;18:17539447241253134. doi: 10.1177/17539447241253134. Ther Adv Cardiovasc Dis. 2024. PMID: 38819836 Free PMC article. Review.
Cited by
-
Resveratrol Attenuate Myocardial Injury by Inhibiting Ferroptosis Via Inducing KAT5/GPX4 in Myocardial Infarction.Front Pharmacol. 2022 May 24;13:906073. doi: 10.3389/fphar.2022.906073. eCollection 2022. Front Pharmacol. 2022. PMID: 35685642 Free PMC article.
-
MicroRNA-195-5p Downregulation Inhibits Endothelial Mesenchymal Transition and Myocardial Fibrosis in Diabetic Cardiomyopathy by Targeting Smad7 and Inhibiting Transforming Growth Factor Beta 1-Smads-Snail Pathway.Front Physiol. 2021 Sep 30;12:709123. doi: 10.3389/fphys.2021.709123. eCollection 2021. Front Physiol. 2021. PMID: 34658906 Free PMC article.
-
Serum microRNAs in Systemic Sclerosis, Associations with Digital Vasculopathy and Lung Involvement.Int J Mol Sci. 2022 Sep 14;23(18):10731. doi: 10.3390/ijms231810731. Int J Mol Sci. 2022. PMID: 36142646 Free PMC article.
-
miR-129-5p restores cardiac function in rats with chronic heart failure by targeting the E3 ubiquitin ligase Smurf1 and promoting PTEN expression.Bioengineered. 2022 Feb;13(2):2371-2386. doi: 10.1080/21655979.2021.2024335. Bioengineered. 2022. PMID: 35034538 Free PMC article.
-
Post-myocardial infarction fibrosis: Pathophysiology, examination, and intervention.Front Pharmacol. 2023 Mar 28;14:1070973. doi: 10.3389/fphar.2023.1070973. eCollection 2023. Front Pharmacol. 2023. PMID: 37056987 Free PMC article. Review.
References
-
- Dickstein K., Cohen-Solal A., Filippatos G. et al. . (2008) ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur. Heart J. 29, 2388–2442 - PubMed
-
- Hunt S.A., Abraham W.T., Chin M.H. et al. . (2009) 2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the International Society for Heart and Lung Transplantation. J. Am. Coll. Cardiol. 53, e1–e90 - PubMed
-
- Hamaguchi S., Tsuchihashi-Makaya M., Kinugawa S. et al. . (2009) Chronic kidney disease as an independent risk for long-term adverse outcomes in patients hospitalized with heart failure in Japan. Report from the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD). Circ. J. 73, 1442–1447 10.1253/circj.CJ-09-0062 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

