Non-neutralizing antibody responses following A(H1N1)pdm09 influenza vaccination with or without AS03 adjuvant system
- PMID: 32889792
- PMCID: PMC7767944
- DOI: 10.1111/irv.12780
Non-neutralizing antibody responses following A(H1N1)pdm09 influenza vaccination with or without AS03 adjuvant system
Abstract
Background: Non-neutralizing antibodies inducing complement-dependent lysis (CDL) and antibody-dependent cell-mediated cytotoxicity (ADCC) activity may contribute to protection against influenza infection. We investigated CDL and ADCC responses in healthy adults randomized to receive either non-adjuvanted or AS03-adjuvanted monovalent A(H1N1)pdm09 vaccine (containing 15 µg/3.75 μg of hemagglutinin, respectively) on a 2-dose schedule 21 days apart.
Methods: We conducted an exploratory analysis of a subset of 106 subjects having no prior history of A(H1N1)pdm09 infection or seasonal influenza vaccination enrolled in a previously reported study (NCT00985673). Antibody responses against the homologous A/California/7/2009 (H1N1) vaccine strain and a related A/Brisbane/59/2007 (H1N1) seasonal influenza strain were analyzed up to Day 42.
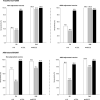
Results: Baseline seropositivity determined with hemagglutination inhibition (HI), CDL and ADCC antibody titers against viral strains was high; A/California/7/2009 (HI [40.4-48.1%]; CDL [34.6-36.0%]; ADCC [92.1-92.3%]); A/Brisbane/59/2007 (HI [73.1-88.9%]; CDL [38.0-42.0%]; ADCC [86.8-97.0%]). CDL seropositivity increased following vaccination with both adjuvanted and non-adjuvanted formulations (A/California/7/2009 [95.9-100%]; A/Brisbane/59/2007 [75.5-79.6%]). At Day 21, increases in CDL and ADCC antibody geometric mean titers against both strains were observed for both formulations. After 2 doses of AS03-adjuvanted vaccine, vaccine responses of 95.8% (≥9-fold increase from baseline in CDL titers) and 34.3% (≥16-fold increase from baseline in ADCC titers) were seen against A/California/7/2009; and 22.4% and 42.9%, respectively, against A/Brisbane/59/2007. Vaccine responses after 2 doses of the non-adjuvanted vaccine were broadly similar.
Conclusions: Broadly comparable non-neutralizing immune responses were observed following vaccination with non-adjuvanted and AS03-adjuvanted A(H1N1)pdm09 formulations; including activity against a related vaccine strain.
Keywords: A(H1N1)pdm09 vaccine; AS03 adjuvant system; cross-reactivity; non-neutralizing antibodies.
© 2020 Glaxosmithkline Biologicals. Influenza and Other Respiratory Viruses Published by John Wiley & Sons Ltd.
Conflict of interest statement
DF, TO, BS, AS and KW are employed by the GSK group of companies. PL and DV were employees of the GSK group of companies during the conduct of the study. BS, AS, KW and PL hold shares from the GSK group of companies. MC and FE received funds from GSK through their institution for the conduct of the study. None of the authors declare non‐financial conflicts of interest.
Figures


References
-
- Kreijtz JH, Fouchier RA, Rimmelzwaan GF. Immune responses to influenza virus infection. Virus Res. 2011;162:19‐30. - PubMed
-
- Kim H, Webster RG, Webby RJ. Influenza Virus: Dealing with a Drifting and Shifting Pathogen. Viral Immunol. 2018;31:174‐183. - PubMed
-
- Hashimoto G, Wright PF, Karzon DT. Antibody‐dependent cell‐mediated cytotoxicity against influenza virus‐infected cells. J Infect Dis. 1983;148:785‐794. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

