Enantioselective Total Synthesis of (-)-Finerenone Using Asymmetric Transfer Hydrogenation
- PMID: 32890415
- PMCID: PMC7839499
- DOI: 10.1002/anie.202011256
Enantioselective Total Synthesis of (-)-Finerenone Using Asymmetric Transfer Hydrogenation
Abstract
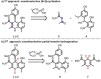
(-)-Finerenone is a nonsteroidal mineralocorticoid receptor antagonist currently in phase III clinical trials for the treatment of chronic kidney disease in type 2 diabetes. It contains an unusual dihydronaphthyridine core. We report a 6-step synthesis of (-)-finerenone, which features an enantioselective partial transfer hydrogenation of a naphthyridine using a chiral phosphoric acid catalyst with a Hantzsch ester. The process is complicated by the fact that the naphthyridine exists as a mixture of two atropisomers that react at different rates and with different selectivities. The intrinsic kinetic resolution was converted into a kinetic dynamic resolution at elevated temperature, which enabled us to obtain (-)-finerenone in both high yield and high enantioselectivity. DFT calculations have revealed the origin of selectivity.
Keywords: MR antagonists; enantioselective synthesis; partial transfer hydrogenation; pharmaceutical molecule.
© 2020 Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- None
-
- Pitt B., Zannad F., Remme W. J., Cody R., Castaigne A., Perez A., Palensky J., Wittes J., N. Engl. J. Med. 1999, 341, 709–717; - PubMed
-
- Pitt B., Remme W., Zannad F., Neaton J., Martinez F., Roniker B., Bittman R., Hurley S., Kleiman J., Gatlin M., N. Engl. J. Med. 2003, 348, 1309–1321; - PubMed
-
- Zannad F., McMurray J. J. V., Krum H., van Veldhuisen D. J., Swedberg K., Shi H., Vincent J., Pocock S. J., Pitt B., N. Engl. J. Med. 2011, 364, 11–21. - PubMed
-
- Gerisch M., Heinig R., Engelen A., Lang D., Kolkhof P., Radtke M., Platzek J., Lovis K., Rohde G., Schwarz T., Drug Metab. Dispos. 2018, 46, 1546–1555. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

