CNS control of the endocrine pancreas
- PMID: 32894319
- PMCID: PMC7983553
- DOI: 10.1007/s00125-020-05204-6
CNS control of the endocrine pancreas
Abstract
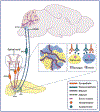
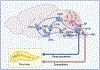
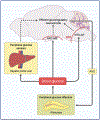
Increasing evidence suggests that, although pancreatic islets can function autonomously to detect and respond to changes in the circulating glucose level, the brain cooperates with the islet to maintain glycaemic control. Here, we review the role of the central and autonomic nervous systems in the control of the endocrine pancreas, including mechanisms whereby the brain senses circulating blood glucose levels. We also examine whether dysfunction in these systems might contribute to complications of type 1 diabetes and the pathogenesis of type 2 diabetes. Graphical abstract.
Keywords: Autonomic nervous system; Brain; Diabetes; Glucagon; Glucose; Insulin; Islet; Pancreas; Review.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

