Suptavumab for the Prevention of Medically Attended Respiratory Syncytial Virus Infection in Preterm Infants
- PMID: 32897368
- PMCID: PMC8653633
- DOI: 10.1093/cid/ciaa951
Suptavumab for the Prevention of Medically Attended Respiratory Syncytial Virus Infection in Preterm Infants
Abstract
Background: Respiratory syncytial virus (RSV) is a major cause of childhood medically attended respiratory infection (MARI).
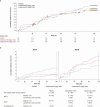
Methods: We conducted a randomized, double-blind, placebo-controlled phase 3 trial in 1154 preterm infants of 1 or 2 doses of suptavumab, a human monoclonal antibody that can bind and block a conserved epitope on RSV A and B subtypes, for the prevention of RSV MARI. The primary endpoint was proportion of subjects with RSV-confirmed hospitalizations or outpatient lower respiratory tract infection (LRTI).
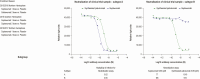
Results: There were no significant differences between primary endpoint rates (8.1%, placebo; 7.7%, 1-dose; 9.3%, 2-dose). Suptavumab prevented RSV A infections (relative risks, .38; 95% confidence interval [CI], .14-1.05 in the 1-dose group and .39 [95% CI, .14-1.07] in the 2-dose group; nominal significance of combined suptavumab group vs placebo; P = .0499), while increasing the rate of RSV B infections (relative risk 1.36 [95% CI, .73-2.56] in the 1-dose group and 1.69 [95% CI, .92-3.08] in the 2-dose group; nominal significance of combined suptavumab group vs placebo; P = .12). Sequenced RSV isolates demonstrated no suptavumab epitope changes in RSV A isolates, while all RSV B isolates had 2-amino acid substitution in the suptavumab epitope that led to loss of neutralization activity. Treatment emergent adverse events were balanced across treatment groups.
Conclusions: Suptavumab did not reduce overall RSV hospitalizations or outpatient LRTI because of a newly circulating mutant strain of RSV B. Genetic variation in circulating RSV strains will continue to challenge prevention efforts.
Clinical trials registration: NCT02325791.
Keywords: efficacy; infants; respiratory syncytial virus; safety.
© The Author(s) 2020. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures

Comment in
-
How Viral Sequence Analysis May Guide Development of Respiratory Syncytial Virus Monoclonal Antibodies.Clin Infect Dis. 2021 Dec 6;73(11):e4409-e4410. doi: 10.1093/cid/ciaa944. Clin Infect Dis. 2021. PMID: 32640025 No abstract available.
References
-
- Mazur NI, Higgins D, Nunes MC, et al. ; Respiratory Syncytial Virus Network (ReSViNET) Foundation . The respiratory syncytial virus vaccine landscape: lessons from the graveyard and promising candidates. Lancet Infect Dis 2018; 18:e295–311. - PubMed
-
- IMpact-RSV. Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from respiratory syncytial virus infection in high-risk infants. Pediatrics 1998; 102:531–7. - PubMed
-
- American Academy of Pediatrics Committee on Infectious Diseases; American Academy of Pediatrics Bronchiolitis Guidelines Committee. Updated guidance for palivizumab prophylaxis among infants and young children at increased risk of hospitalization for respiratory syncytial virus infection. Pediatrics 2014; 134:e620–38. - PubMed
-
- Adis Insight. Suptavumab. Available at: https://adisinsight.springer.com/drugs/800040470. Accessed 5 March 2019.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

