Gut Microbiota between Environment and Genetic Background in Familial Mediterranean Fever (FMF)
- PMID: 32899315
- PMCID: PMC7563178
- DOI: 10.3390/genes11091041
Gut Microbiota between Environment and Genetic Background in Familial Mediterranean Fever (FMF)
Abstract
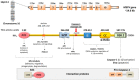
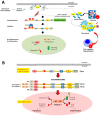
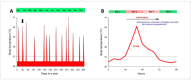
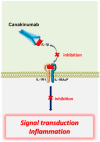
The gastrointestinal tract hosts the natural reservoir of microbiota since birth. The microbiota includes various bacteria that establish a progressively mutual relationship with the host. Of note, the composition of gut microbiota is rather individual-specific and, normally, depends on both the host genotype and environmental factors. The study of the bacterial profile in the gut demonstrates that dominant and minor phyla are present in the gastrointestinal tract with bacterial density gradually increasing in oro-aboral direction. The cross-talk between bacteria and host within the gut strongly contributes to the host metabolism, to structural and protective functions. Dysbiosis can develop following aging, diseases, inflammatory status, and antibiotic therapy. Growing evidences show a possible link between the microbiota and Familial Mediterranean Fever (FMF), through a shift of the relative abundance in microbial species. To which extent such perturbations of the microbiota are relevant in driving the phenotypic manifestations of FMF with respect to genetic background, remains to be further investigated.
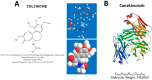
Keywords: MEFV; amyloidosis; colchicine; inflammasome; interleukin-1b.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Predominant role of host genetics in controlling the composition of gut microbiota.PLoS One. 2008 Aug 26;3(8):e3064. doi: 10.1371/journal.pone.0003064. PLoS One. 2008. PMID: 18725973 Free PMC article.
-
Gut microbiota alterations are associated with phenotype and genotype in familial Mediterranean fever.Rheumatology (Oxford). 2024 Apr 2;63(4):1039-1048. doi: 10.1093/rheumatology/kead322. Rheumatology (Oxford). 2024. PMID: 37402619
-
Specific changes in faecal microbiota are associated with familial Mediterranean fever.Ann Rheum Dis. 2019 Oct;78(10):1398-1404. doi: 10.1136/annrheumdis-2019-215258. Epub 2019 Aug 3. Ann Rheum Dis. 2019. PMID: 31377728
-
Familial Mediterranean fever at the millennium. Clinical spectrum, ancient mutations, and a survey of 100 American referrals to the National Institutes of Health.Medicine (Baltimore). 1998 Jul;77(4):268-97. doi: 10.1097/00005792-199807000-00005. Medicine (Baltimore). 1998. PMID: 9715731 Review.
-
Familial Mediterranean fever.Pediatr Nephrol. 2003 Sep;18(9):853-9. doi: 10.1007/s00467-003-1185-2. Epub 2003 Jun 27. Pediatr Nephrol. 2003. PMID: 12836090 Review.
Cited by
-
Placebo-resistant gut bacteria: Akkermansia muciniphila spp. and Familial Mediterranean fever disease.Front Cell Infect Microbiol. 2024 Feb 23;14:1336752. doi: 10.3389/fcimb.2024.1336752. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 38465231 Free PMC article. Clinical Trial.
-
Nutraceuticals and pharmacological to balance the transitional microbiome to extend immunity during COVID-19 and other viral infections.J Transl Med. 2024 Sep 18;22(1):847. doi: 10.1186/s12967-024-05587-9. J Transl Med. 2024. PMID: 39294611 Free PMC article. Review.
-
Analysis of the fecal and oral microbiota in chronic recurrent multifocal osteomyelitis.Arthritis Res Ther. 2022 Feb 22;24(1):54. doi: 10.1186/s13075-021-02711-8. Arthritis Res Ther. 2022. PMID: 35193655 Free PMC article.
-
Gut Microbiota Dysbiosis Correlates with Abnormal Immune Response in Moderate COVID-19 Patients with Fever.J Inflamm Res. 2021 Jun 17;14:2619-2631. doi: 10.2147/JIR.S311518. eCollection 2021. J Inflamm Res. 2021. PMID: 34168484 Free PMC article.
-
Anticancer Mechanism of Astragalus Polysaccharide and Its Application in Cancer Immunotherapy.Pharmaceuticals (Basel). 2024 May 15;17(5):636. doi: 10.3390/ph17050636. Pharmaceuticals (Basel). 2024. PMID: 38794206 Free PMC article. Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

