Potential Role of PDGFRβ-Associated THBS4 in Colorectal Cancer Development
- PMID: 32899998
- PMCID: PMC7564555
- DOI: 10.3390/cancers12092533
Potential Role of PDGFRβ-Associated THBS4 in Colorectal Cancer Development
Abstract
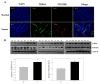
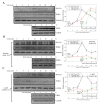
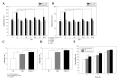
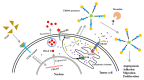
Colorectal cancer is a significant cause of death since it frequently metastasizes to several organs such as the lung or liver. Tumor development is affected by various factors, including a tumor microenvironment, which may be an essential factor that leads to tumor growth, proliferation, invasion, and metastasis. In the tumor microenvironment, abnormal changes in various growth factors, enzymes, and cytokines can wield a strong influence on cancer. Thrombospondin-4 (THBS4), which is an extracellular matrix protein, also plays essential roles in the tumor microenvironment and mediates angiogenesis by transforming growth factor-β (TGFβ) signaling. Platelet-derived growth factor receptor β (PDGFRβ), which is a receptor tyrosine kinase and is also a downstream signal of TGFβ, is associated with invasion and metastasis in colorectal cancer. We identified that PDGFRβ and THBS4 are overexpressed in tumor tissues of colorectal cancer patients, and that PDGF-D expression increased after TGFβ treatment in the colon cancer cell line DLD-1. TGFβ and PDGF-D increased cellular THBS4 protein levels and secretion but did not increase THBS4 mRNA levels. This response was further confirmed by the inositol 1,4,5-triphosphate receptor (IP3R) and stromal interaction molecule 1 (STIM1) blockade as well as the PDGFRβ blockade. We propose that the PDGFRβ signal leads to a modification of the incomplete form of THBS4 to its complete form through IP3R, STIM1, and Ca2+-signal proteins, which further induces THBS4 secretion. Additionally, we identified that DLD-1 cell-conditioned medium stimulated with PDGF-D promotes adhesion, migration, and proliferation of colon myofibroblast CCD-18co cells, and this effect was intensified in the presence of thrombin. These findings suggest that excessive PDGFRβ signaling due to increased TGFβ and PDGF-D in colorectal tumors leads to over-secretion of THBS4 and proliferative tumor development.
Keywords: Ca2+; PDGFRβ; THBS4; colorectal cancer.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results
Figures



Similar articles
-
Complex formation between platelet-derived growth factor receptor β and transforming growth factor β receptor regulates the differentiation of mesenchymal stem cells into cancer-associated fibroblasts.Oncotarget. 2018 Sep 25;9(75):34090-34102. doi: 10.18632/oncotarget.26124. eCollection 2018 Sep 25. Oncotarget. 2018. PMID: 30344924 Free PMC article.
-
PDGF-D/PDGFRβ promotes tongue squamous carcinoma cell (TSCC) progression via activating p38/AKT/ERK/EMT signal pathway.Biochem Biophys Res Commun. 2016 Sep 16;478(2):845-51. doi: 10.1016/j.bbrc.2016.08.035. Epub 2016 Aug 6. Biochem Biophys Res Commun. 2016. PMID: 27507215
-
Over-expression of Thrombospondin 4 correlates with loss of miR-142 and contributes to migration and vascular invasion of advanced hepatocellular carcinoma.Oncotarget. 2017 Apr 4;8(14):23277-23288. doi: 10.18632/oncotarget.15054. Oncotarget. 2017. PMID: 28177895 Free PMC article.
-
Diverse roles of tumor-stromal PDGFB-to-PDGFRβ signaling in breast cancer growth and metastasis.Adv Cancer Res. 2022;154:93-140. doi: 10.1016/bs.acr.2022.01.003. Epub 2022 Feb 24. Adv Cancer Res. 2022. PMID: 35459473 Review.
-
The role of small molecule platelet-derived growth factor receptor (PDGFR) inhibitors in the treatment of neoplastic disorders.Pharmacol Res. 2018 Mar;129:65-83. doi: 10.1016/j.phrs.2018.01.021. Epub 2018 Feb 3. Pharmacol Res. 2018. PMID: 29408302 Review.
Cited by
-
Machine Learning Predicts the Oxidative Stress Subtypes Provide an Innovative Insight into Colorectal Cancer.Oxid Med Cell Longev. 2023 Apr 21;2023:1737501. doi: 10.1155/2023/1737501. eCollection 2023. Oxid Med Cell Longev. 2023. PMID: 37122535 Free PMC article.
-
Signaling networks in cancer stromal senescent cells establish malignant microenvironment.Proc Natl Acad Sci U S A. 2025 Apr 8;122(14):e2412818122. doi: 10.1073/pnas.2412818122. Epub 2025 Apr 1. Proc Natl Acad Sci U S A. 2025. PMID: 40168129
-
A simplified and efficient extracellular vesicle-based proteomics strategy for early diagnosis of colorectal cancer.Chem Sci. 2024 Oct 15;15(44):18419-30. doi: 10.1039/d4sc05518g. Online ahead of print. Chem Sci. 2024. PMID: 39421202 Free PMC article.
-
Identification hub genes of consensus molecular subtype correlation with immune infiltration and predict prognosis in gastric cancer.Discov Oncol. 2021 Oct 16;12(1):41. doi: 10.1007/s12672-021-00434-5. Discov Oncol. 2021. PMID: 35201473 Free PMC article.
-
Pathological Significance and Prognostic Roles of Thrombospondin-3, 4 and 5 in Bladder Cancer.In Vivo. 2021 May-Jun;35(3):1693-1701. doi: 10.21873/invivo.12429. In Vivo. 2021. PMID: 33910854 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

