Engineered high-affinity zinc binding site reveals gating configurations of a human proton channel
- PMID: 32902579
- PMCID: PMC7537347
- DOI: 10.1085/jgp.202012664
Engineered high-affinity zinc binding site reveals gating configurations of a human proton channel
Abstract
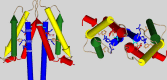
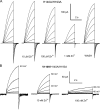
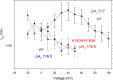
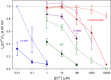
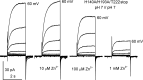
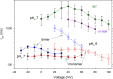
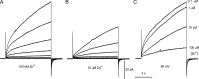
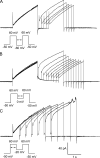
The voltage-gated proton channel (HV1) is a voltage sensor that also conducts protons. The singular ability of protons to penetrate proteins complicates distinguishing closed and open channels. When we replaced valine with histidine at position 116 in the external vestibule of hHV1, current was potently inhibited by externally applied Zn2+ in a construct lacking the two His that bind Zn2+ in WT channels. High-affinity binding with profound effects at 10 nM Zn2+ at pHo 7 suggests additional groups contribute. We hypothesized that Asp185, which faces position 116 in our closed-state model, contributes to Zn2+ chelation. Confirming this prediction, V116H/D185N abolished Zn2+ binding. Studied in a C-terminal truncated monomeric construct, V116H channels activated rapidly. Anomalously, Zn2+ slowed activation, producing a time constant independent of both voltage and Zn2+ concentration. We hypothesized that slow turn-on of H+ current in the presence of Zn2+ reflects the rate of Zn2+ unbinding from the channel, analogous to drug-receptor dissociation reactions. This behavior in turn suggests that the affinity for Zn2+ is greater in the closed state of hHV1. Supporting this hypothesis, pulse pairs revealed a rapid component of activation whose amplitude decreased after longer intervals at negative voltages as closed channels bound Zn2+. The lower affinity of Zn2+ in open channels is consistent with the idea that structural rearrangements within the transmembrane region bring Arg205 near position 116, electrostatically expelling Zn2+. This phenomenon provides direct evidence that Asp185 opposes position 116 in closed channels and that Arg205 moves between them when the channel opens.
© 2020 Cherny et al.
Figures



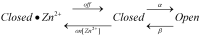
Comment in
-
Zn2+ to probe voltage-gated proton (Hv1) channels.J Gen Physiol. 2020 Oct 5;152(10):e202012725. doi: 10.1085/jgp.202012725. J Gen Physiol. 2020. PMID: 32910187 Free PMC article.
References
-
- Banh R., Cherny V.V., Morgan D., Musset B., Thomas S., Kulleperuma K., Smith S.M.E., Pomès R., and DeCoursey T.E.. 2019. Hydrophobic gasket mutation produces gating pore currents in closed human voltage-gated proton channels. Proc. Natl. Acad. Sci. USA. 116:18951–18961. 10.1073/pnas.1905462116 - DOI - PMC - PubMed

