Facile H/D Exchange at (Hetero)Aromatic Hydrocarbons Catalyzed by a Stable Trans-Dihydride N-Heterocyclic Carbene (NHC) Iron Complex
- PMID: 32902969
- PMCID: PMC7586338
- DOI: 10.1021/jacs.0c07689
Facile H/D Exchange at (Hetero)Aromatic Hydrocarbons Catalyzed by a Stable Trans-Dihydride N-Heterocyclic Carbene (NHC) Iron Complex
Abstract
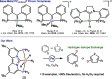
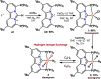
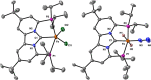
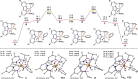
Earth-abundant metal pincer complexes have played an important role in homogeneous catalysis during the last ten years. Yet, despite intense research efforts, the synthesis of iron PCcarbeneP pincer complexes has so far remained elusive. Here we report the synthesis of the first PCNHCP functionalized iron complex [(PCNHCP)FeCl2] (1) and the reactivity of the corresponding trans-dihydride iron(II) dinitrogen complex [(PCNHCP)Fe(H)2N2)] (2). Complex 2 is stable under an atmosphere of N2 and is highly active for hydrogen isotope exchange at (hetero)aromatic hydrocarbons under mild conditions (50 °C, N2). With benzene-d6 as the deuterium source, easily reducible functional groups such as esters and amides are well tolerated, contributing to the overall wide substrate scope (e.g., halides, ethers, and amines). DFT studies suggest a complex assisted σ-bond metathesis pathway for C(sp2)-H bond activation, which is further discussed in this study.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
-
- Cornils B.; Herrmann W. A.; Beller M.; Paciello R.. Applied Homogeneous Catalysis with Organometallic Compounds: A Comprehensive Handbook in Four Vol., 3rd ed.; Wiley-VCH Verlag GmbH & Co: Weinheim, Germany, 2017.
- Hartwig J. F.Organotransition Metal Chemistry: From Bonding to Catalysis; University Science Books: Sausalito, CA, 2010.
-
- Hapke M.; Hilt G.. Cobalt Catalysis in Organic Synthesis: Methods and Reactions; Wiley-VCH Verlag GmbH & Co: Weinheim, Germany, 2020.
-
- Fürstner A. Iron Catalysis in Organic Synthesis: A Critical Assessment of What It Takes To Make This Base Metal a Multitasking Champion. ACS Cent. Sci. 2016, 2, 778–789. 10.1021/acscentsci.6b00272. - DOI - PMC - PubMed
- Bauer I.; Knölker H.-J. Iron Catalysis in Organic Synthesis. Chem. Rev. 2015, 115, 3170–3387. 10.1021/cr500425u. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

