Complement Activation by C-Reactive Protein Is Critical for Protection of Mice Against Pneumococcal Infection
- PMID: 32903624
- PMCID: PMC7438579
- DOI: 10.3389/fimmu.2020.01812
Complement Activation by C-Reactive Protein Is Critical for Protection of Mice Against Pneumococcal Infection
Abstract
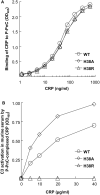
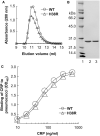
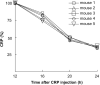
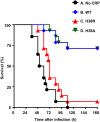
C-reactive protein (CRP), a component of the innate immune system, is an antipneumococcal plasma protein. Human CRP has been shown to protect mice against infection with lethal doses of Streptococcus pneumoniae by decreasing bacteremia. in vitro, CRP binds to phosphocholine-containing substances, such as pneumococcal C-polysaccharide, in a Ca2+-dependent manner. Phosphocholine-complexed human CRP activates the complement system in both human and murine sera. The mechanism of antipneumococcal action of CRP in vivo, however, has not been defined yet. In this study, we tested a decades-old hypothesis that the complement-activating property of phosphocholine-complexed CRP contributes to protection of mice against pneumococcal infection. Our approach was to investigate a CRP mutant, incapable of activating murine complement, in mouse protection experiments. We employed site-directed mutagenesis of CRP, guided by its three-dimensional structure, and identified a mutant H38R which, unlike wild-type CRP, did not activate complement in murine serum. Substitution of His38 with Arg in CRP did not affect the pentameric structure of CRP, did not affect the binding of CRP to pneumococci, and did not decrease the stability of CRP in mouse circulation. Employing a murine model of pneumococcal infection, we found that passively administered H38R CRP failed to protect mice against infection. Infected mice injected with H38R CRP showed no reduction in bacteremia and did not survive longer, as opposed to infected mice treated with wild-type CRP. Thus, the hypothesis that complement activation by phosphocholine-complexed CRP is an antipneumococcal effector function was supported. We can conclude now that complement activation by phosphocholine-complexed CRP is indeed essential for CRP-mediated protection of mice against pneumococcal infection.
Keywords: C-reactive protein; acute phase response; complement; inflammation; pneumococcal infection.
Copyright © 2020 Singh, Ngwa and Agrawal.
Figures

Similar articles
-
Protection against prolonged pneumococcal infection involves structural changes in C-reactive protein and subsequent binding to both phosphocholine and amyloids on the bacterial surface.Front Immunol. 2025 Jul 16;16:1631409. doi: 10.3389/fimmu.2025.1631409. eCollection 2025. Front Immunol. 2025. PMID: 40740789 Free PMC article.
-
The phosphocholine-binding pocket on C-reactive protein is necessary for initial protection of mice against pneumococcal infection.J Biol Chem. 2012 Dec 14;287(51):43116-25. doi: 10.1074/jbc.M112.427310. Epub 2012 Nov 8. J Biol Chem. 2012. PMID: 23139417 Free PMC article.
-
C-reactive protein protects mice against pneumococcal infection via both phosphocholine-dependent and phosphocholine-independent mechanisms.Infect Immun. 2015 May;83(5):1845-52. doi: 10.1128/IAI.03058-14. Epub 2015 Feb 17. Infect Immun. 2015. PMID: 25690104 Free PMC article.
-
Structure-Function Relationships of C-Reactive Protein in Bacterial Infection.Front Immunol. 2019 Feb 26;10:166. doi: 10.3389/fimmu.2019.00166. eCollection 2019. Front Immunol. 2019. PMID: 30863393 Free PMC article. Review.
-
The protective function of human C-reactive protein in mouse models of Streptococcus pneumoniae infection.Endocr Metab Immune Disord Drug Targets. 2008 Dec;8(4):231-7. doi: 10.2174/187153008786848321. Endocr Metab Immune Disord Drug Targets. 2008. PMID: 19075776 Free PMC article. Review.
Cited by
-
An evolutionarily conserved function of C-reactive protein is to prevent the formation of amyloid fibrils.Front Immunol. 2024 Sep 16;15:1466865. doi: 10.3389/fimmu.2024.1466865. eCollection 2024. Front Immunol. 2024. PMID: 39351235 Free PMC article.
-
Protection against prolonged pneumococcal infection involves structural changes in C-reactive protein and subsequent binding to both phosphocholine and amyloids on the bacterial surface.Front Immunol. 2025 Jul 16;16:1631409. doi: 10.3389/fimmu.2025.1631409. eCollection 2025. Front Immunol. 2025. PMID: 40740789 Free PMC article.
-
C-reactive protein: structure, function, regulation, and role in clinical diseases.Front Immunol. 2024 Jun 14;15:1425168. doi: 10.3389/fimmu.2024.1425168. eCollection 2024. Front Immunol. 2024. PMID: 38947332 Free PMC article. Review.
-
C-Reactive Protein-Based Strategy to Reduce Antibiotic Dosing for the Treatment of Pneumococcal Infection.Front Immunol. 2021 Jan 20;11:620784. doi: 10.3389/fimmu.2020.620784. eCollection 2020. Front Immunol. 2021. PMID: 33552084 Free PMC article.
-
Treatment of Pneumococcal Infection by Using Engineered Human C-Reactive Protein in a Mouse Model.Front Immunol. 2020 Oct 7;11:586669. doi: 10.3389/fimmu.2020.586669. eCollection 2020. Front Immunol. 2020. PMID: 33117400 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

