Orthohantavirus Pathogenesis and Cell Tropism
- PMID: 32903721
- PMCID: PMC7438779
- DOI: 10.3389/fcimb.2020.00399
Orthohantavirus Pathogenesis and Cell Tropism
Abstract
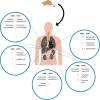
Orthohantaviruses are zoonotic viruses that are naturally maintained by persistent infection in specific reservoir species. Although these viruses mainly circulate among rodents worldwide, spill-over infection to humans occurs. Orthohantavirus infection in humans can result in two distinct clinical outcomes: hemorrhagic fever with renal syndrome (HFRS) and hantavirus cardiopulmonary syndrome (HCPS). While both syndromes develop following respiratory transmission and are associated with multi-organ failure and high mortality rates, little is known about the mechanisms that result in these distinct clinical outcomes. Therefore, it is important to identify which cell types and tissues play a role in the differential development of pathogenesis in humans. Here, we review current knowledge on cell tropism and its role in pathogenesis during orthohantavirus infection in humans and reservoir rodents. Orthohantaviruses predominantly infect microvascular endothelial cells (ECs) of a variety of organs (lungs, heart, kidney, liver, and spleen) in humans. However, in this review we demonstrate that other cell types (e.g., macrophages, dendritic cells, and tubular epithelium) are infected as well and may play a role in the early steps in pathogenesis. A key driver for pathogenesis is increased vascular permeability, which can be direct effect of viral infection in ECs or result of an imbalanced immune response in an attempt to clear the virus. Future studies should focus on the role of identifying how infection of organ-specific endothelial cells as well as other cell types contribute to pathogenesis.
Keywords: endothelium; hantavirus; hantavirus cardiopulmonary syndrome; hemorrhagic fever with renal syndrome; orthohantavirus; pathogenesis; tropism.
Copyright © 2020 Noack, Goeijenbier, Reusken, Koopmans and Rockx.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

