Mitochondrial Contact Sites in Inflammation-Induced Cardiovascular Disease
- PMID: 32903766
- PMCID: PMC7438832
- DOI: 10.3389/fcell.2020.00692
Mitochondrial Contact Sites in Inflammation-Induced Cardiovascular Disease
Abstract
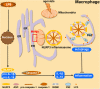
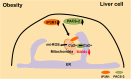
The mitochondrion, the ATP-producing center, is both physically and functionally associated with almost all other organelles in the cell. Mitochondrial-associated membranes (MAMs) are involved in a variety of biological processes, such as lipid exchange, protein transport, mitochondrial fission, mitophagy, and inflammation. Several inflammation-related diseases in the cardiovascular system involve several intracellular events including mitochondrial dysfunction as well as disruption of MAMs. Therefore, an in-depth exploration of the function of MAMs will be of great significance for us to understand the initiation, progression, and clinical complications of cardiovascular disease (CVD). In this review, we summarize the recent advances in our knowledge of MAM regulation and function in CVD-related cells. We discuss the potential roles of MAMs in activating inflammation to influence the development of CVD.
Keywords: autophagy; cardiovascular disease; inflammasome; inflammation; mitochondria; mitochondrial-associated membranes.
Copyright © 2020 Liu, Liu, Zhuang, Fan, Zhu, Xu, He, Liu and Feng.
Figures







Similar articles
-
Mitochondria-Associated Endoplasmic Reticulum Membranes in Cardiovascular Diseases.Front Cell Dev Biol. 2020 Nov 9;8:604240. doi: 10.3389/fcell.2020.604240. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33240899 Free PMC article. Review.
-
Structure and Function of Mitochondria-Associated Endoplasmic Reticulum Membranes (MAMs) and Their Role in Cardiovascular Diseases.Oxid Med Cell Longev. 2021 Jul 11;2021:4578809. doi: 10.1155/2021/4578809. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 34336092 Free PMC article. Review.
-
Hyperglycemia-Driven Inhibition of AMP-Activated Protein Kinase α2 Induces Diabetic Cardiomyopathy by Promoting Mitochondria-Associated Endoplasmic Reticulum Membranes In Vivo.Circulation. 2019 Apr 16;139(16):1913-1936. doi: 10.1161/CIRCULATIONAHA.118.033552. Circulation. 2019. PMID: 30646747 Free PMC article.
-
Targeting mitochondria-associated membranes as a potential therapy against endothelial injury induced by hypoxia.J Cell Biochem. 2019 Nov;120(11):18967-18978. doi: 10.1002/jcb.29220. Epub 2019 Jun 26. J Cell Biochem. 2019. PMID: 31241212
-
Three-dimensional organization of the endoplasmic reticulum membrane around the mitochondrial constriction site in mammalian cells revealed by using focused-ion beam tomography.Microscopy (Oxf). 2014 Nov;63 Suppl 1:i34. doi: 10.1093/jmicro/dfu076. Microscopy (Oxf). 2014. PMID: 25359839
Cited by
-
Mitochondria inter-organelle relationships in cancer protein aggregation.Front Cell Dev Biol. 2022 Dec 19;10:1062993. doi: 10.3389/fcell.2022.1062993. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36601538 Free PMC article. Review.
-
Cardiovascular protection of YiyiFuzi powder and the potential mechanisms through modulating mitochondria-endoplasmic reticulum interactions.Front Pharmacol. 2024 Jun 24;15:1405545. doi: 10.3389/fphar.2024.1405545. eCollection 2024. Front Pharmacol. 2024. PMID: 38978978 Free PMC article. Review.
-
The MFN2 Q367H variant reveals a novel pathomechanism connected to mtDNA-mediated inflammation.Life Sci Alliance. 2025 Apr 2;8(6):e202402921. doi: 10.26508/lsa.202402921. Print 2025 Jun. Life Sci Alliance. 2025. PMID: 40175090 Free PMC article.
-
Electrophysiological Mechanisms and Therapeutic Potential of Calcium Channels in Atrial Fibrillation.Rev Cardiovasc Med. 2025 Jun 25;26(6):33507. doi: 10.31083/RCM33507. eCollection 2025 Jun. Rev Cardiovasc Med. 2025. PMID: 40630446 Free PMC article. Review.
-
Targeting Mediators of Inflammation in Heart Failure: A Short Synthesis of Experimental and Clinical Results.Int J Mol Sci. 2021 Dec 2;22(23):13053. doi: 10.3390/ijms222313053. Int J Mol Sci. 2021. PMID: 34884857 Free PMC article. Review.
References
-
- Bhaskar V., Yin J., Mirza A. M., Phan D., Vanegas S., Issafras H., et al. (2011). Monoclonal antibodies targeting IL-1 beta reduce biomarkers of atherosclerosis in vitro and inhibit atherosclerotic plaque formation in Apolipoprotein E-deficient mice. Atherosclerosis 216 313–320. 10.1016/j.atherosclerosis.2011.02.026 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

