Synthesis of UV-11 MOF and Its Characterization by Cyclic Voltammetry
- PMID: 32903784
- PMCID: PMC7438935
- DOI: 10.3389/fchem.2020.00617
Synthesis of UV-11 MOF and Its Characterization by Cyclic Voltammetry
Abstract
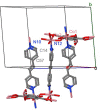
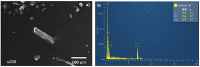
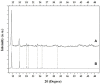
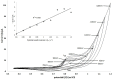
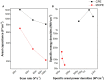
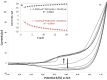
In this work a Metal-Organic Framework (MOF) was prepared using a solvothermal method, taking as precursors 1. 2-di-(4-pyridyl)-ethylene, 1.2.4.5-benzenetetracarboxylic acid and Co(No3)2-6H2O. This MOF was called UV-11 and was evaluated using microscopic, spectroscopic and electrochemical techniques. According to the obtained results, the melting point of the compound is located in a higher interval than its precursors. Stereoscopic microscopy analysis shows the presence of pink crystals in the form of needles. MEB technique displays a laminar morphology as well as crystals with approximate sizes (36 mm wide and 150 mm long). EDS analysis corroborated the presence of precursor elements such as cobalt, carbon and oxygen. Furthermore, the XRD technique shows the cobalt-related phases in the sample, which is cobalt bis (pyridine-6-carboxylic-2-carboxylate). A modified carbon paste electrode was prepared using MOF UV-11 and by cyclic voltammetry electrochemical technique, semi-reversible redox processes are identified, as well as thermodynamic and kinetic parameters were obtained with the Laviron equation, and electrochemical performance properties from the cyclic voltammetry experimental data.
Keywords: MOF (Metal–Organic Framework); characterization-analysis; electroanalysis; electrochemistry (cyclic voltametry); interface mechanical behavior; micro supercapacitor.
Copyright © 2020 Meza-Pardo, Morales-Tapia, Veloz Rodríguez, Reyes-Cruz, Perez-Labra, Urbano-Reyes, Rivera-Villanueva and Cobos-Murcia.
Figures



References
-
- Alfonso-Herrera L. A., Huerta-Flores A. M., Torres-Martínez L. M., Rivera-Villanueva J. M., Ramírez-Herrera D. J. (2018). Hybrid SrZrO3-MOF heterostructure: surface assembly and photocatalytic performance for hydrogen evolution and degradation of indigo carmine dye. J. Mater. Sci. Mater. Electron. 29, 10395–10410. 10.1007/s10854-018-9096-y - DOI
-
- Betteridge W. (1980). The properties of metallic cobalt. Prog. Mater. Sci. 24, 51–142. 10.1016/0079-6425(79)90004-5 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

