BRG1 Activates PR65A Transcription to Regulate NO Bioavailability in Vascular Endothelial Cells
- PMID: 32903816
- PMCID: PMC7443572
- DOI: 10.3389/fcell.2020.00774
BRG1 Activates PR65A Transcription to Regulate NO Bioavailability in Vascular Endothelial Cells
Abstract
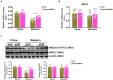
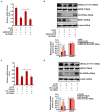
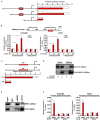
Vascular endothelial cells contribute to the pathogenesis of cardiovascular diseases by producing and disseminating angiocrine factors. Nitric oxide (NO), catalyzed by endothelial NO synthase (eNOS), is one of the prototypical angiocrine factors. eNOS activity is modulated by site-specific phosphorylation. We have previously shown that endothelial-specific knockdown of BRG1 in Apoe -/- mice attenuates the development of atherosclerosis, in which eNOS-dependent NO catalysis plays an antagonizing role. Here we report that attenuation of atherogenesis in mice by BRG1 knockdown was accompanied by partial restoration of NO biosynthesis by 44% in the arteries and a simultaneous up-regulation of eNOS serine 1177 phosphorylation by 59%. Indeed, BRG1 depletion or inhibition ameliorated oxLDL-induced loss of NO bioavailability and eNOS phosphorylation in cultured endothelial cells. Further analysis revealed that BRG1 regulated eNOS phosphorylation and NO synthesis by activating the transcription of protein phosphatase 2A (PP2A) structural subunit a (encoded by PR65A). BRG1 interacted with ETS1, was recruited by ETS1 to the PR65A promoter, and cooperated with ETS1 to activate PR65A transcription. Finally, depletion of ETS1, similar to BRG1, repressed PR65A induction, normalized eNOS phosphorylation, and rescued NO biosynthesis in endothelial cells treated with oxLDL. In conclusion, our data characterize a novel transcriptional cascade that regulates NO bioavailability in vascular endothelial cells.
Keywords: BRG1; PP2A; eNOS; nitric oxide; phosphatase; phosphorylation; transcriptional regulation; vascular endothelial cells.
Copyright © 2020 Chen, Zhao, Xu, Yu, Zhuo, Yang and Xu.
Figures



Similar articles
-
Transcriptional Activation of Matricellular Protein Spondin2 (SPON2) by BRG1 in Vascular Endothelial Cells Promotes Macrophage Chemotaxis.Front Cell Dev Biol. 2020 Aug 14;8:794. doi: 10.3389/fcell.2020.00794. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32974343 Free PMC article.
-
BRG1 deficiency in endothelial cells alleviates thioacetamide induced liver fibrosis in mice.Biochem Biophys Res Commun. 2020 Jan 1;521(1):212-219. doi: 10.1016/j.bbrc.2019.10.109. Epub 2019 Oct 18. Biochem Biophys Res Commun. 2020. PMID: 31635808
-
B56α subunit of protein phosphatase 2A mediates retinoic acid-induced decreases in phosphorylation of endothelial nitric oxide synthase at serine 1179 and nitric oxide production in bovine aortic endothelial cells.Biochem Biophys Res Commun. 2013 Jan 11;430(2):476-81. doi: 10.1016/j.bbrc.2012.12.011. Epub 2012 Dec 10. Biochem Biophys Res Commun. 2013. PMID: 23237802
-
Site-specific dephosphorylation of endothelial nitric oxide synthase by protein phosphatase 2A: evidence for crosstalk between phosphorylation sites.Biochemistry. 2002 Dec 31;41(52):15845-53. doi: 10.1021/bi026732g. Biochemistry. 2002. PMID: 12501214
-
Regulation of endothelial nitric oxide synthase activity and gene expression.Ann N Y Acad Sci. 2002 May;962:122-30. doi: 10.1111/j.1749-6632.2002.tb04062.x. Ann N Y Acad Sci. 2002. PMID: 12076969 Review.
Cited by
-
The Chromatin Remodeling Protein BRG1 Regulates SREBP Maturation by Activating SCAP Transcription in Hepatocytes.Front Cell Dev Biol. 2021 Feb 25;9:622866. doi: 10.3389/fcell.2021.622866. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33718362 Free PMC article.
-
SIRT6 mediates MRTF-A deacetylation in vascular endothelial cells to antagonize oxLDL-induced ICAM-1 transcription.Cell Death Discov. 2022 Mar 4;8(1):96. doi: 10.1038/s41420-022-00903-y. Cell Death Discov. 2022. PMID: 35246513 Free PMC article.
-
BRG1 Deficiency Promotes Cardiomyocyte Inflammation and Apoptosis by Activating the cGAS-STING Signaling in Diabetic Cardiomyopathy.Inflammation. 2025 Feb;48(1):299-315. doi: 10.1007/s10753-024-02058-7. Epub 2024 Jun 13. Inflammation. 2025. PMID: 38867118 Free PMC article.
-
Trans-activation of eotaxin-1 by Brg1 contributes to liver regeneration.Cell Death Dis. 2022 May 25;13(5):495. doi: 10.1038/s41419-022-04944-0. Cell Death Dis. 2022. PMID: 35614068 Free PMC article.
-
Dual Regulation of Tank Binding Kinase 1 by BRG1 in Hepatocytes Contributes to Reactive Oxygen Species Production.Front Cell Dev Biol. 2021 Oct 1;9:745985. doi: 10.3389/fcell.2021.745985. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34660604 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

