Developments in the Application of Nanomaterials for Water Treatment and Their Impact on the Environment
- PMID: 32906594
- PMCID: PMC7558965
- DOI: 10.3390/nano10091764
Developments in the Application of Nanomaterials for Water Treatment and Their Impact on the Environment
Abstract
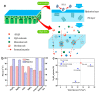
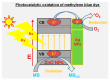
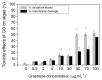
Nanotechnology is an uppermost priority area of research in several nations presently because of its enormous capability and financial impact. One of the most promising environmental utilizations of nanotechnology has been in water treatment and remediation where various nanomaterials can purify water by means of several mechanisms inclusive of the adsorption of dyes, heavy metals, and other pollutants, inactivation and removal of pathogens, and conversion of harmful materials into less harmful compounds. To achieve this, nanomaterials have been generated in several shapes, integrated to form different composites and functionalized with active components. Additionally, the nanomaterials have been added to membranes that can assist to improve the water treatment efficiency. In this paper, we have discussed the advantages of nanomaterials in applications such as adsorbents (removal of dyes, heavy metals, pharmaceuticals, and organic contaminants from water), membrane materials, catalytic utilization, and microbial decontamination. We discuss the different carbon-based nanomaterials (carbon nanotubes, graphene, graphene oxide, fullerenes, etc.), and metal and metal-oxide based nanomaterials (zinc-oxide, titanium dioxide, nano zerovalent iron, etc.) for the water treatment application. It can be noted that the nanomaterials have the ability for improving the environmental remediation system. The examination of different studies confirmed that out of the various nanomaterials, graphene and its derivatives (e.g., reduced graphene oxide, graphene oxide, graphene-based metals, and graphene-based metal oxides) with huge surface area and increased purity, outstanding environmental compatibility and selectivity, display high absorption capability as they trap electrons, avoiding their recombination. Additionally, we discussed the negative impacts of nanomaterials such as membrane damage and cell damage to the living beings in the aqueous environment. Acknowledgment of the possible benefits and inadvertent hazards of nanomaterials to the environment is important for pursuing their future advancement.
Keywords: adsorbents; environment; membranes; nanomaterials; toxicity; water pollution.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Capek I. Nanocomposite Structures and Dispersions: Science and Nanotechnology—Fundamental Principles and Colloidal Particles Studies in Interface Science. In: Mobius D., Miller R., editors. Nanotechnology and Nanomaterials. Volume 23. Elsevier; Amsterdam, The Netherlands: 2006. Chapter 1. - DOI
-
- Chaturvedi S., Dave P.N. Handbook of Nanomaterials for Industrial Applications. Elsevier; Amsterdam, The Netherlands: 2018. Nanomaterials: Environmental, Human Health Risk; pp. 1055–1062. - DOI
-
- Baruah S., Khan M.N., Dutta J. Perspectives and applications of nanotechnology in water treatment. Environ. Chem. Lett. 2016;14:1–14. doi: 10.1007/s10311-015-0542-2. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

