Loss of ADAM9 Leads to Modifications of the Extracellular Matrix Modulating Tumor Growth
- PMID: 32906814
- PMCID: PMC7564588
- DOI: 10.3390/biom10091290
Loss of ADAM9 Leads to Modifications of the Extracellular Matrix Modulating Tumor Growth
Abstract
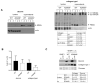
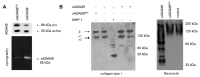
ADAM9 is a metalloproteinase strongly expressed at the tumor-stroma border by both tumor and stromal cells. We previously showed that the host deletion of ADAM9 leads to enhanced growth of grafted B16F1 melanoma cells by a mechanism mediated by TIMP1 and the TNF-α/sTNFR1 pathway. This study aimed to dissect the structural modifications in the tumor microenvironment due to the stromal expression of ADAM9 during melanoma progression. We performed proteomic analysis of peritumoral areas of ADAM9 deleted mice and identified the altered expression of several matrix proteins. These include decorin, collagen type XIV, fibronectin, and collagen type I. Analysis of these matrices in the matrix producing cells of the dermis, fibroblasts, showed that ADAM9-/- and wild type fibroblasts synthesize and secreted almost comparable amounts of decorin. Conversely, collagen type I expression was moderately, but not significantly, decreased at the transcriptional level, and the protein increased in ADAM9-/- fibroblast mono- and co-cultures with melanoma media. We show here for the first time that ADAM9 can release a collagen fragment. Still, it is not able to degrade collagen type I. However, the deletion of ADAM9 in fibroblasts resulted in reduced MMP-13 and -14 expression that may account for the reduced processing of collagen type I. Altogether, the data show that the ablation of ADAM9 in the host leads to the altered expression of peritumoral extracellular matrix proteins that generate a more favorable environment for melanoma cell growth. These data underscore the suppressive role of stromal expression of ADAM9 in tumor growth and call for a better understanding of how protease activities function in a cellular context for improved targeting.
Keywords: ADAM9; collagen type I; extracellular matrix; fibroblasts; melanoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

