Childhood Langerhans cell histiocytosis with severe lung involvement: a nationwide cohort study
- PMID: 32907615
- PMCID: PMC7487928
- DOI: 10.1186/s13023-020-01495-5
Childhood Langerhans cell histiocytosis with severe lung involvement: a nationwide cohort study
Abstract
Background: Lung involvement in childhood Langerhans cell histiocytosis (LCH) is infrequent and rarely life threatening, but occasionally, severe presentations are observed.
Methods: Among 1482 children (< 15 years) registered in the French LCH registry (1994-2018), 111 (7.4%) had lung involvement. This retrospective study included data for 17 (1.1%) patients that required one or more intensive care unit (ICU) admissions for respiratory failure.
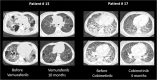
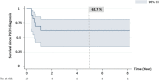
Results: The median age was 1.3 years at the first ICU hospitalization. Of the 17 patients, 14 presented with lung involvement at the LCH diagnosis, and 7 patients (41%) had concomitant involvement of risk-organ (hematologic, spleen, or liver). Thirty-five ICU hospitalizations were analysed. Among these, 22 (63%) were secondary to a pneumothorax, 5 (14%) were associated with important cystic lesions without pneumothorax, and 8 (23%) included a diffuse micronodular lung infiltration in the context of multisystem disease. First-line vinblastine-corticosteroid combination therapy was administered to 16 patients; 12 patients required a second-line therapy (cladribine: n = 7; etoposide-aracytine: n = 3; targeted therapy n = 2). A total of 6 children (35%) died (repeated pneumothorax: n = 3; diffuse micronodular lung infiltration in the context of multisystem disease: n = 2; following lung transplantation: n = 1). For survivors, the median follow-up after ICU was 11.2 years. Among these, 9 patients remain asymptomatic despite abnormal chest imaging.
Conclusions: Severe lung involvement is unusual in childhood LCH, but it is associated with high mortality. Treatment guidelines should be improved for this group of patients: viral infection prophylaxis and early administration of a new LCH therapy, such as targeted therapy.
Keywords: Childhood; Intensive care; Langerhans cell histiocytosis; Pulmonary; Severe; Targeted therapy.
Conflict of interest statement
The French LCH registry was supported by a grant from Roche. J.D. received travel support and honoraria from X4 Pharma. J.-F.E. received honoraria from Bristol-Myers Squibb, MSD Oncology, HalioDx, Pierre Fabre, and Amgen. We declare no other conflict of interest.
Figures


References
-
- Guyot-Goubin A, Donadieu J, Barkaoui M, Bellec S, Thomas C, Clavel J. Descriptive epidemiology of childhood Langerhans cell histiocytosis in France, 2000–2004. Pediatr Blood Cancer. 2008;51:71–75. - PubMed
-
- Donadieu J, Guyot-Goubin A, Clavel J, Thomas C. Présentation clinique et épidémiologie de l’histiocytose langerhansienne chez l’enfant. Arch Pediatr. 2008;15:520–522. - PubMed
-
- Ronceray L, Pötschger U, Janka G, Gadner H, Minkov M. Pulmonary Involvement in Pediatric-Onset Multisystem Langerhans Cell Histiocytosis: Effect on Course and Outcome. J Pediatrics. 2012;161:129–133.e3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

