Neural progenitor cell pyroptosis contributes to Zika virus-induced brain atrophy and represents a therapeutic target
- PMID: 32907937
- PMCID: PMC7519283
- DOI: 10.1073/pnas.2007773117
Neural progenitor cell pyroptosis contributes to Zika virus-induced brain atrophy and represents a therapeutic target
Erratum in
-
Correction to Supporting Information for He et al., Neural progenitor cell pyroptosis contributes to Zika virus-induced brain atrophy and represents a therapeutic target.Proc Natl Acad Sci U S A. 2021 Dec 7;118(49):e2119180118. doi: 10.1073/pnas.2119180118. Proc Natl Acad Sci U S A. 2021. PMID: 34819337 Free PMC article. No abstract available.
Abstract
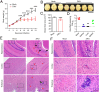
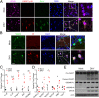
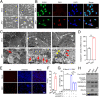
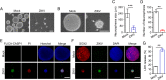
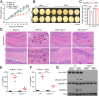
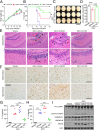
Mounting evidence has associated Zika virus (ZIKV) infection with congenital malformations, including microcephaly, which raises global alarm. Nonetheless, mechanisms by which ZIKV disrupts neurogenesis and causes microcephaly are far from being understood. In this study, we discovered direct effects of ZIKV on neural progenitor cell development by inducing caspase-1- and gasdermin D (GSDMD)-mediated pyroptotic cell death, linking ZIKV infection with the development of microcephaly. Importantly, caspase-1 depletion or its inhibitor VX-765 treatment reduced ZIKV-induced inflammatory responses and pyroptosis, and substantially attenuated neuropathology and brain atrophy in vivo. Collectively, our data identify caspase-1- and GSDMD-mediated pyroptosis in neural progenitor cells as a previously unrecognized mechanism for ZIKV-related pathological effects during neural development, and also provide treatment options for ZIKV-associated diseases.
Keywords: Zika virus; caspase-1; microcephaly; neurogenesis; pyroptosis.
Conflict of interest statement
The authors declare no competing interest.
Figures
References
-
- Dick G. W., Zika virus. II. Pathogenicity and physical properties. Trans. R. Soc. Trop. Med. Hyg. 46, 521–534 (1952). - PubMed
-
- World Health Organization , Zika epidemiology update. https://www.who.int/emergencies/diseases/zika/epidemiology-update/en/. Accessed 2 July 2019.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

