Vascular dimorphism ensured by regulated proteoglycan dynamics favors rapid umbilical artery closure at birth
- PMID: 32909945
- PMCID: PMC7529456
- DOI: 10.7554/eLife.60683
Vascular dimorphism ensured by regulated proteoglycan dynamics favors rapid umbilical artery closure at birth
Abstract
The umbilical artery lumen closes rapidly at birth, preventing neonatal blood loss, whereas the umbilical vein remains patent longer. Here, analysis of umbilical cords from humans and other mammals identified differential arterial-venous proteoglycan dynamics as a determinant of these contrasting vascular responses. The umbilical artery, but not the vein, has an inner layer enriched in the hydrated proteoglycan aggrecan, external to which lie contraction-primed smooth muscle cells (SMC). At birth, SMC contraction drives inner layer buckling and centripetal displacement to occlude the arterial lumen, a mechanism revealed by biomechanical observations and confirmed by computational analyses. This vascular dimorphism arises from spatially regulated proteoglycan expression and breakdown. Mice lacking aggrecan or the metalloprotease ADAMTS1, which degrades proteoglycans, demonstrate their opposing roles in umbilical vascular dimorphism, including effects on SMC differentiation. Umbilical vessel dimorphism is conserved in mammals, suggesting that differential proteoglycan dynamics and inner layer buckling were positively selected during evolution.
Keywords: birth; computational biology; developmental biology; extracellular matrix; human; mouse; proteoglycans; systems biology; umbilical cord; vascular engineering; vascular smooth muscle.
© 2020, Nandadasa et al.
Conflict of interest statement
SN, JS, VP, SM, CK, AO, CN, CH, BC, MD, NS, KT, MV, DS, EP, JH, SA No competing interests declared
Figures
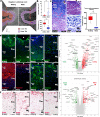


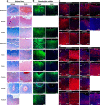

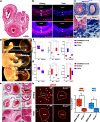
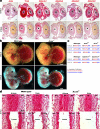
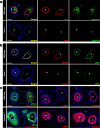
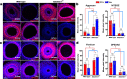


Comment in
-
Severing umbilical ties.Elife. 2020 Oct 12;9:e63128. doi: 10.7554/eLife.63128. Elife. 2020. PMID: 33044167 Free PMC article.
References
-
- Baek S, Gleason RL, Rajagopal KR, Humphrey JD. Theory of small on large: potential utility in computations of fluid–solid interactions in arteries. Computer Methods in Applied Mechanics and Engineering. 2007;196:3070–3078. doi: 10.1016/j.cma.2006.06.018. - DOI
-
- Demirkoparan H, Pence TJ. Swelling of an internally pressurized nonlinearly elastic tube with fiber reinforcing. International Journal of Solids and Structures. 2007;44:4009–4029. doi: 10.1016/j.ijsolstr.2006.11.006. - DOI
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

