Intrauterine Vacuum-Induced Hemorrhage-Control Device for Rapid Treatment of Postpartum Hemorrhage
- PMID: 32909970
- PMCID: PMC7575019
- DOI: 10.1097/AOG.0000000000004138
Intrauterine Vacuum-Induced Hemorrhage-Control Device for Rapid Treatment of Postpartum Hemorrhage
Abstract
Objective: To evaluate the effectiveness and safety of an intrauterine vacuum-induced hemorrhage-control device for postpartum hemorrhage treatment.
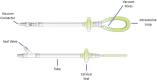
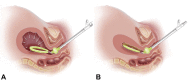
Methods: A multicenter, prospective, single-arm treatment study of a novel intrauterine device that uses low-level vacuum to induce uterine myometrial contraction to achieve control of abnormal postpartum uterine bleeding and postpartum hemorrhage was undertaken at 12 centers in the United States. The primary effectiveness endpoint was the proportion of participants in whom use of the intrauterine vacuum-induced hemorrhage-control device controlled abnormal bleeding without requiring escalating interventions. The primary safety endpoint was the incidence, severity, and seriousness of device-related adverse events. Secondary outcomes included time to bleeding control, rate of transfusion, and device usability scored by each investigator using the device.
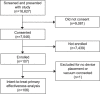
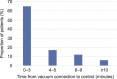
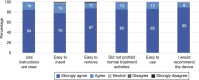
Results: Of 107 participants enrolled with primary postpartum hemorrhage or abnormal postpartum uterine bleeding, 106 received any study treatment with the device connected to vacuum, and successful treatment was observed in 94% (100/106, 95% CI 88-98%) of these participants. In those 100 participants, definitive control of abnormal bleeding was reported in a median of 3 minutes (interquartile range 2.0-5.0) after connection to vacuum. Eight adverse events deemed possibly related to the device or procedure were reported, all of which were outlined as risks in the study and all of which resolved with treatment without serious clinical sequelae. Transfusion of 1-3 units of red blood cells was required in 35 participants, and five participants required 4 or more units of red blood cells. The majority of investigators reported the intrauterine vacuum-induced hemorrhage-control device as easy to use (98%) and would recommend it (97%).
Conclusion: Intrauterine vacuum-induced hemorrhage control may provide a new rapid and effective treatment option for abnormal postpartum uterine bleeding or postpartum hemorrhage, with the potential to prevent severe maternal morbidity and mortality.
Funding source: Alydia Health, Inc.
Clinical trial registration: ClinicalTrials.gov, NCT02883673.
Figures
Comment in
-
New Approaches to the Age-Old Problem of Uterine Atony.Obstet Gynecol. 2020 Nov;136(5):874-875. doi: 10.1097/AOG.0000000000004156. Obstet Gynecol. 2020. PMID: 33095541 No abstract available.
-
Intrauterine Vacuum-Induced Hemorrhage-Control Device for Rapid Treatment of Postpartum Hemorrhage.Obstet Gynecol. 2021 Jun 1;137(6):1127. doi: 10.1097/AOG.0000000000004418. Obstet Gynecol. 2021. PMID: 34011877 No abstract available.
-
In Reply.Obstet Gynecol. 2021 Jun 1;137(6):1127-1128. doi: 10.1097/AOG.0000000000004419. Obstet Gynecol. 2021. PMID: 34011878 No abstract available.
References
-
- Gülmezoglu AM, Lawrie TA, Hezelgrave N, Oladapo OT, Souza JP, Gielen M, et al. Interventions to reduce maternal and newborn morbidity and mortality. In: Black RE, Laxminarayan R, Temmerman M, Walker N, editors. Reproductive, maternal, newborn, and child health: disease control priorities, 3rd ed Washington, DC: The International Bank for Reconstruction and Development/The World Bank; 2016. - PubMed
-
- Bateman BT, Berman MF, Riley LE, Leffert LR. The epidemiology of postpartum hemorrhage in a large, nationwide sample of deliveries. Anesthesia Analgesia 2010;110:1368–73. - PubMed
-
- Goffman D, Friedman AM, Sheen JJ, Kessler A, Vawdrey D, Green R, et al. A framework for improving characterization of obstetric hemorrhage using informatics data. Obstet Gynecol 2019;134:1317–25. - PubMed
-
- Main EK, Goffman D, Scavone BM, Low LK, Bingham D, Fontaine PL, et al. National partnership for maternal safety, Obstet Gynecol 2015;126:155–62. - PubMed
-
- Dena G, Ananth CV, Fleischer A, D'Alton M, Lavery JA, Smiley R, et al. The New York State Safe Motherhood Initiative: early impact of obstetric hemorrhage bundle implementation. Am J Perinatology 2019;36:1344–50. - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

