Feasibility of Long-term Proteasome Inhibition in Multiple Myeloma by in-class Transition From Bortezomib to Ixazomib
- PMID: 32912820
- PMCID: PMC7336931
- DOI: 10.1016/j.clml.2020.06.024
Feasibility of Long-term Proteasome Inhibition in Multiple Myeloma by in-class Transition From Bortezomib to Ixazomib
Abstract
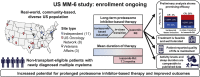
Background: The ongoing US MM-6 study is investigating in-class transition (iCT) from parenteral bortezomib-based induction to all-oral IRd (ixazomib-lenalidomide-dexamethasone) with the aim of increasing proteasome inhibitor (PI)-based treatment adherence and duration while maintaining patients' health-related quality of life (HRQoL) and improving outcomes.
Patients and methods: US community sites are enrolling non-transplant-eligible patients with newly diagnosed multiple myeloma (MM) with no evidence of progressive disease after 3 cycles of bortezomib-based therapy to receive IRd (up to 39 cycles or until progression or toxicity). The patients use mobile or wearable digital devices to collect actigraphy (activity and sleep) data and electronically complete HRQoL, treatment satisfaction and medication adherence questionnaires. The primary endpoint is progression-free survival. The key secondary endpoints include response rates and therapy duration.
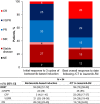
Results: At the data cutoff, 84 patients had been treated (median age 73 years; 44% aged ≥ 75 years; 49% men; 15% Black or African American; and 10% Hispanic or Latino). Of the 84 patients, 62% were continuing therapy. The mean duration of total PI therapy was 10.1 months and for the IRd regimen was 7.3 months. With an 8-month median follow-up, the 12-month progression-free survival rate was 86% (95% confidence interval, 73%-93%) from both the start of bortezomib-based treatment and the start of IRd. The overall response rate was 62% (complete response, 4%; very good partial response, 25%; partial response, 33%) after bortezomib-based induction and 70% (complete response, 26%; very good partial response, 29%; partial response, 15%) after iCT. The IRd safety profile was consistent with previous clinical trial data, and HRQoL and treatment satisfaction were maintained.
Conclusion: The patients included in the US MM-6 study are representative of the real-world US MM population. The use of iCT might permit prolonged PI-based therapy with promising efficacy, without impacting patients' HRQoL or treatment satisfaction.
Keywords: Duration of treatment; Medication adherence; Oral therapy; Patient-reported outcomes; Real-world community.
Copyright © 2020 The Authors. Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Assessment of prolonged proteasome inhibition through ixazomib-based oral regimen on newly diagnosed and first-relapsed multiple myeloma: A real-world Chinese cohort study.Cancer Med. 2024 May;13(9):e7177. doi: 10.1002/cam4.7177. Cancer Med. 2024. PMID: 38686615 Free PMC article.
-
Patient Characteristics and Outcomes of Relapsed/Refractory Multiple Myeloma in Patients Treated with Proteasome Inhibitors in Germany.Oncol Res Treat. 2020;43(9):449-459. doi: 10.1159/000509018. Epub 2020 Jul 21. Oncol Res Treat. 2020. PMID: 32694243 Free PMC article.
-
Survival benefit of ixazomib, lenalidomide and dexamethasone (IRD) over lenalidomide and dexamethasone (Rd) in relapsed and refractory multiple myeloma patients in routine clinical practice.BMC Cancer. 2021 Jan 15;21(1):73. doi: 10.1186/s12885-020-07732-1. BMC Cancer. 2021. PMID: 33451293 Free PMC article.
-
Ixazomib - the first oral proteasome inhibitor.Leuk Lymphoma. 2019 Mar;60(3):610-618. doi: 10.1080/10428194.2018.1523398. Epub 2019 Jan 7. Leuk Lymphoma. 2019. PMID: 30614337 Review.
-
Ixazomib for the treatment of multiple myeloma.Expert Opin Investig Drugs. 2015;24(9):1287-98. doi: 10.1517/13543784.2015.1065250. Epub 2015 Jul 3. Expert Opin Investig Drugs. 2015. PMID: 26138345 Review.
Cited by
-
Assessment of prolonged proteasome inhibition through ixazomib-based oral regimen on newly diagnosed and first-relapsed multiple myeloma: A real-world Chinese cohort study.Cancer Med. 2024 May;13(9):e7177. doi: 10.1002/cam4.7177. Cancer Med. 2024. PMID: 38686615 Free PMC article.
-
Application of Patient-Generated Health Data Among Older Adults With Cancer: Scoping Review.J Med Internet Res. 2025 Feb 4;27:e57379. doi: 10.2196/57379. J Med Internet Res. 2025. PMID: 39903506 Free PMC article.
-
In-class transition (iCT) of proteasome inhibitor-based therapy: a community approach to multiple myeloma management.Blood Cancer J. 2023 Sep 19;13(1):147. doi: 10.1038/s41408-023-00912-9. Blood Cancer J. 2023. PMID: 37726298 Free PMC article.
-
The acceptability of using wearable electronic devices to monitor physical activity of patients with Multiple Myeloma undergoing treatment: a systematic review.Clin Hematol Int. 2024 Jul 29;6(3):38-53. doi: 10.46989/001c.121406. eCollection 2024. Clin Hematol Int. 2024. PMID: 39268172 Free PMC article.
-
Efficacy and Safety of Ixazomib Plus Lenalidomide and Dexamethasone Following Injectable PI-Based Therapy in Relapsed/Refractory Multiple Myeloma.Ann Hematol. 2023 Sep;102(9):2493-2504. doi: 10.1007/s00277-023-05212-7. Epub 2023 Jun 21. Ann Hematol. 2023. PMID: 37341778 Free PMC article.
References
-
- Kumar S.K., Callander N.S., Hillengass J., et al. NCCN guidelines insights: multiple myeloma, version 1.2020. J Natl Compr Canc Netw. 2019;17:1154–1165. - PubMed
-
- Durie B.G.M., Hoering A., Abidi M.H., et al. Bortezomib with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma without intent for immediate autologous stem-cell transplant (SWOG S0777): a randomised, open-label, phase 3 trial. Lancet. 2017;389:519–527. - PMC - PubMed
-
- San Miguel J.F., Schlag R., Khuageva N.K., et al. Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N Engl J Med. 2008;359:906–917. - PubMed
-
- San Miguel J.F., Schlag R., Khuageva N.K., et al. Persistent overall survival benefit and no increased risk of second malignancies with bortezomib-melphalan-prednisone versus melphalan-prednisone in patients with previously untreated multiple myeloma. J Clin Oncol. 2013;31:448–455. - PubMed
-
- Mateos M.V., Richardson P.G., Dimopoulos M.A., et al. Effect of cumulative bortezomib dose on survival in multiple myeloma patients receiving bortezomib-melphalan-prednisone in the phase III VISTA study. Am J Hematol. 2015;90:314–319. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

