CoQ10 suppression of oxidative stress and cell senescence increases bone mass in orchiectomized mice
- PMID: 32913507
- PMCID: PMC7476121
CoQ10 suppression of oxidative stress and cell senescence increases bone mass in orchiectomized mice
Erratum in
-
Erratum: CoQ10 suppression of oxidative stress and cell senescence increases bone mass in orchiectomized mice.Am J Transl Res. 2021 Apr 15;13(4):3923. eCollection 2021. Am J Transl Res. 2021. PMID: 34017585 Free PMC article.
Abstract
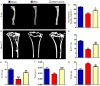
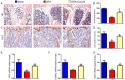
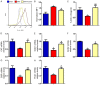
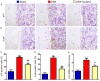
Numerous studies support the detrimental effects of oxidative stress and cell senescence on skeletal homeostasis. Coenzyme Q10 (CoQ10) acts as a scavenger for oxidative stress and protects mitochondrial activity from oxidative damage. However, it is unclear whether CoQ10 has a protective effect on osteoporosis caused by orchiectomy. To investigate suppression effect of antioxidant CoQ10 on osteoporosis in orchiectomized (ORX) mice, ORX mice were supplemented with/without CoQ10, and were compared with each other and with sham-operated mice. Our results showed that CoQ10 could prevent ORX-induced bone loss by inhibiting oxidative stress and cell senescence, subsequently promoting osteoblastic bone formation and inhibiting osteoclastic bone resorption. The results of this study not only reveal the mechanism of CoQ10 supplementation in anti-osteoporosis, but also provide experimental and theoretical basis for the clinical application of CoQ10 in the prevention of osteoporosis.
Keywords: ORX-induced bone loss; cell senescence; coenzyme Q10; oxidative stress.
AJTR Copyright © 2020.
Conflict of interest statement
None.
Figures


Similar articles
-
N-acetylcysteine prevents orchiectomy-induced osteoporosis by inhibiting oxidative stress and osteocyte senescence.Am J Transl Res. 2019 Jul 15;11(7):4337-4347. eCollection 2019. Am J Transl Res. 2019. PMID: 31396339 Free PMC article.
-
Pyrroloquinoline quinone prevents testosterone deficiency-induced osteoporosis by stimulating osteoblastic bone formation and inhibiting osteoclastic bone resorption.Am J Transl Res. 2017 Mar 15;9(3):1230-1242. eCollection 2017. Am J Transl Res. 2017. PMID: 28386349 Free PMC article.
-
Astaxanthin attenuates irradiation-induced osteoporosis in mice by inhibiting oxidative stress, osteocyte senescence, and SASP.Food Funct. 2022 Nov 14;13(22):11770-11779. doi: 10.1039/d2fo01673g. Food Funct. 2022. PMID: 36285709
-
Clinical Evidence for Q10 Coenzyme Supplementation in Heart Failure: From Energetics to Functional Improvement.J Clin Med. 2020 Apr 27;9(5):1266. doi: 10.3390/jcm9051266. J Clin Med. 2020. PMID: 32349341 Free PMC article. Review.
-
Therapeutic Potential of Co-enzyme Q10 in Retinal Diseases.Curr Med Chem. 2017;24(39):4329-4339. doi: 10.2174/0929867324666170801100516. Curr Med Chem. 2017. PMID: 28762311 Review.
Cited by
-
Effect of coenzyme Q10 on tibial fracture resistance in nicotine-exposed rats.PLoS One. 2025 Jan 3;20(1):e0315462. doi: 10.1371/journal.pone.0315462. eCollection 2025. PLoS One. 2025. PMID: 39752356 Free PMC article.
-
The Role of Antioxidants in the Interplay between Oxidative Stress and Senescence.Antioxidants (Basel). 2022 Jun 22;11(7):1224. doi: 10.3390/antiox11071224. Antioxidants (Basel). 2022. PMID: 35883714 Free PMC article. Review.
References
-
- Kanis JA, Melton LJ 3rd, Christiansen C, Johnston CC, Khaltaev N. The diagnosis of osteoporosis. J Bone Miner Res. 1994;9:1137–1141. - PubMed
-
- Resch H, Gollob E, Kudlacek S, Pietschmann P. Osteoporosis in the man. Wien Med Wochenschr. 2001;151:457–463. - PubMed
-
- McCarthy TL, Centrella M. Androgen receptor activation integrates complex transcriptional effects in osteoblasts, involving the growth factors TGF-beta and IGF-I, and transcription factor C/EBPdelta. Gene. 2015;573:129–140. - PubMed
-
- Takano H, Zou Y, Hasegawa H, Akazawa H, Nagai T, Komuro I. Oxidative stress-induced signal transduction pathways in cardiac myocytes: involvement of ROS in heart diseases. Antioxid Redox Signal. 2003;5:789–794. - PubMed
LinkOut - more resources
Full Text Sources
