Mutational Landscape in Resected Periampullary Adenocarcinoma: Relationship With Morphology and Clinical Outcome
- PMID: 32914025
- PMCID: PMC7446458
- DOI: 10.1200/PO.18.00323
Mutational Landscape in Resected Periampullary Adenocarcinoma: Relationship With Morphology and Clinical Outcome
Abstract
Purpose: Periampullary adenocarcinomas encompass a heterogeneous group of tumors with dismal prognosis and limited treatment options. Emerging evidence shows that tumor morphology (ie, intestinal type [I-type] or pancreatobiliary type [PB-type]) is a more relevant prognostic factor than tumor origin. Knowledge is sparse, however, on whether key mutations differ according to morphology.
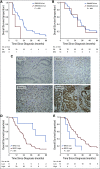
Materials and methods: Next-generation sequencing was applied to assess the mutational status of 70 genes in 102 tumors from a retrospective cohort of 175 patients with resected periampullary adenocarcinoma. Brahma-related gene 1 protein expression was examined by immunohistochemistry on tissue microarrays with primary tumors from the original cohort.
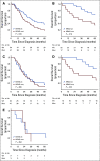
Results: APC mutations were significantly more common in I-type than in PB-type tumors (27.5% v 0%; P < .001), as were ERBB3 mutations (20.8% v 4.8%; P = .016), whereas CDKN2A mutations were more common in PB-type than in I-type tumors (19.4% v 2.5%; P = .013). KRAS mutation was an independent factor of poor prognosis in I-type tumors (hazard ratio, 3.73; 95% CI, 1.10 to 12.67). In PB-type tumors, SMARCA4 mutation was an adverse prognostic factor in patients not receiving adjuvant chemotherapy, and there was a significant treatment interaction between expression of Brahma-related gene 1 protein, the protein encoded by SMARCA4, and adjuvant chemotherapy (P interaction = .007).
Conclusion: To our knowledge, this is the first description of the mutational landscape in the full spectrum of periampullary adenocarcinoma that demonstrates that the distribution and prognostic and predictive significance of commonly mutated genes differ by morphology. The results emphasize that morphology is an important factor to consider in the search for novel biomarkers and targeted personalized treatment of these patients. In addition, the findings support the concept that molecular profiling of these tumors could be of clinical benefit.
© 2019 by American Society of Clinical Oncology.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to www.asco.org/rwc or ascopubs.org/po/author-center. Margareta HebyConsulting or Advisory Role: Bristol-Myers SquibbKarolina HolmEmployment: SAGA Diagnostics No other potential conflicts of interest were reported.
Figures

References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

