Striated Preferentially Expressed Protein Kinase (SPEG)-Deficient Skeletal Muscles Display Fewer Satellite Cells with Reduced Proliferation and Delayed Differentiation
- PMID: 32919980
- PMCID: PMC7734599
- DOI: 10.1016/j.ajpath.2020.08.012
Striated Preferentially Expressed Protein Kinase (SPEG)-Deficient Skeletal Muscles Display Fewer Satellite Cells with Reduced Proliferation and Delayed Differentiation
Abstract
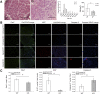
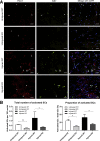
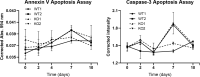
Centronuclear myopathies (CNMs) are a subtype of congenital myopathies characterized by skeletal muscle weakness and an increase in the number of central myonuclei. SPEG (striated preferentially expressed protein kinase) has been identified as the sixth gene associated with CNM, and it has been shown that striated muscle-specific Speg-knockout (KO) mice have defective triad formation, abnormal excitation-contraction coupling, and calcium mishandling. The impact of SPEG deficiency on the survival and function of myogenic cells remains to be deciphered. In this study, the authors examined the overall population, proliferation, and differentiation of myogenic cells obtained from striated muscle-specific Speg-KO mice and compared them with wild-type (WT) controls. SPEG-deficient skeletal muscles contained fewer myogenic cells, which on further study demonstrated reduced proliferation and delayed differentiation compared with those from WT muscles. Regenerative response to skeletal muscle injury in Speg-KO mice was compared with that of WT mice, leading to the identification of similar abnormalities including fewer satellite cells, fewer dividing cells, and an increase in apoptotic cells in KO mice. Overall, these results reveal specific abnormalities in myogenic cell number and behavior associated with SPEG deficiency. Similar satellite cell defects have been reported in mouse models of MTM1- and DNM2-associated CNM, suggestive of shared underlying pathways.
Copyright © 2020 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures







References
-
- Phadke R. Myopathology of congenital myopathies: bridging the old and the new. Semin Pediatr Neurol. 2019;29:55–70. - PubMed
-
- Agrawal P.B., Pierson C.R., Joshi M., Liu X., Ravenscroft G., Moghadaszadeh B., Talabere T., Viola M., Swanson L.C., Haliloglu G., Talim B., Yau K.S., Allcock R.J., Laing N.G., Perrella M.A., Beggs A.H. SPEG interacts with myotubularin, and its deficiency causes centronuclear myopathy with dilated cardiomyopathy. Am J Hum Genet. 2014;95:218–226. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

