Distribution of GFAP in Squamata: Extended Immunonegative Areas, Astrocytes, High Diversity, and Their Bearing on Evolution
- PMID: 32922269
- PMCID: PMC7457009
- DOI: 10.3389/fnana.2020.00049
Distribution of GFAP in Squamata: Extended Immunonegative Areas, Astrocytes, High Diversity, and Their Bearing on Evolution
Abstract
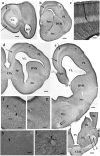
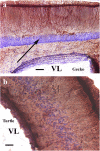
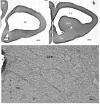
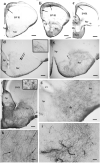
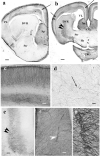
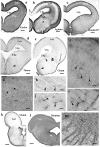
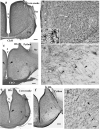
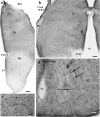
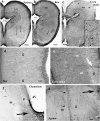
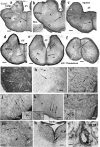
Squamata is one of the richest and most diverse extant groups. The present study investigates the glial fibrillary acidic protein (GFAP)-immunopositive elements of five lizard and three snake species; each represents a different family. The study continues our former studies on bird, turtle, and caiman brains. Although several studies have been published on lizards, they usually only investigated one species. Almost no data are available on snakes. The animals were transcardially perfused. Immunoperoxidase reactions were performed with a mouse monoclonal anti-GFAP (Novocastra). The original radial ependymoglia is enmeshed by secondary, non-radial processes almost beyond recognition in several brain areas like in other reptiles. Astrocytes occur but only as complementary elements like in caiman but unlike in turtles, where astrocytes are absent. In most species, extended areas are free of GFAP-a meaningful difference from other reptiles. The predominance of astrocytes and the presence of areas free of GFAP immunopositivity are characteristic of birds and mammals; therefore, they must be apomorphic features of Squamata, which appeared independently from the evolution of avian glia. However, these features show a high diversity; in some lizards, they are even absent. There was no principal difference between the glial structures of snakes and lizards. In conclusion, the glial structure of Squamata seems to be the most apomorphic one among reptiles. The high diversity suggests that its evolution is still intense. The comparison of identical brain areas with different GFAP contents in different species may promote understanding the role of GFAP in neuronal networks. Our findings are in accordance with the supposal based on our previous studies that the GFAP-free areas expand during evolution.
Keywords: brain evolution; glial fibrillary acidic protein (GFAP); lizards; snakes; turtles.
Copyright © 2020 Lõrincz and Kálmán.
Figures



Similar articles
-
Glial fibrillary acidic protein-immunopositive structures in the brain of a Crocodilian, Caiman crocodilus, and its bearing on the evolution of astroglia.J Comp Neurol. 2001 Mar 19;431(4):460-80. doi: 10.1002/1096-9861(20010319)431:4<460::aid-cne1083>3.3.co;2-8. J Comp Neurol. 2001. PMID: 11223815
-
Distribution of glial fibrillary acidic protein and vimentin-immunopositive elements in the developing chicken brain from hatch to adulthood.Anat Embryol (Berl). 1998 Sep;198(3):213-35. doi: 10.1007/s004290050179. Anat Embryol (Berl). 1998. PMID: 9764976
-
Glial fibrillary acidic protein and vimentin immunoreactivity of astroglial cells in the central nervous system of adult Podarcis sicula (Squamata, Lacertidae).J Anat. 2001 Jan;198(Pt 1):67-75. doi: 10.1046/j.1469-7580.2001.19810067.x. J Anat. 2001. PMID: 11215769 Free PMC article.
-
GFAP-immunopositive structures in spiny dogfish, Squalus acanthias, and little skate, Raia erinacea, brains: differences have evolutionary implications.Anat Embryol (Berl). 2001 Jul;204(1):59-80. doi: 10.1007/s004290100180. Anat Embryol (Berl). 2001. PMID: 11506433
-
GFAP expression withdraws--a trend of glial evolution?Brain Res Bull. 2002 Feb-Mar 1;57(3-4):509-11. doi: 10.1016/s0361-9230(01)00713-4. Brain Res Bull. 2002. PMID: 11923020 Review.
Cited by
-
Evolution of astrocytes: From invertebrates to vertebrates.Front Cell Dev Biol. 2022 Aug 15;10:931311. doi: 10.3389/fcell.2022.931311. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36046339 Free PMC article. Review.
-
The relative withdrawal of GFAP-An essential component of brain evolution.Front Neuroanat. 2025 Jul 17;19:1607603. doi: 10.3389/fnana.2025.1607603. eCollection 2025. Front Neuroanat. 2025. PMID: 40747269 Free PMC article.
-
Evolution of Astrocyte-Neuron Interactions Across Species.Adv Neurobiol. 2024;39:1-17. doi: 10.1007/978-3-031-64839-7_1. Adv Neurobiol. 2024. PMID: 39190069 Review.
-
Astrocyte evolution and human specificity.Neural Regen Res. 2023 Jan;18(1):131-132. doi: 10.4103/1673-5374.340405. Neural Regen Res. 2023. PMID: 35799529 Free PMC article. No abstract available.
-
Cytoskeleton Markers in the Spinal Cord and Mechanoreceptors of Thick-Toed Geckos after Prolonged Space Flights.Life (Basel). 2022 Jan 11;12(1):100. doi: 10.3390/life12010100. Life (Basel). 2022. PMID: 35054493 Free PMC article.
References
-
- Achúcarro N. (1915). De l’évulotion de la névroglie, et spécialement de ses relations avec 1′appareil vasculaire. Trab. Lab. Invest. Biol. 13 169–212.
-
- Appel B. (2013). “Nonmammalian vertebrate glia,” in Neuroglia, 3rd Edn, eds Kettenmann H., Ransom B. R. (New York, NY: Oxford University Press; ), 24–34. 10.1093/med/9780199794591.003.0003 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

