Rapid detection of high-risk HPV16 and HPV18 based on microchip electrophoresis
- PMID: 32923006
- PMCID: PMC7474136
- DOI: 10.1016/j.jpha.2020.04.003
Rapid detection of high-risk HPV16 and HPV18 based on microchip electrophoresis
Abstract
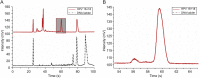
Researches on detection of human papillomavirus (HPV) high-risk samples were carried out by polymerase chain reaction (PCR) coupled with microchip electrophoresis (MCE). Herein, we introduced a simple, rapid, automated method for detecting high-risk samples HPV16 and HPV18. In this research, general primers were initially selected to obtain sufficient detectable yield by PCR to verify feasibility of MCM method for HPV detection, then type-specific primers were further used to evaluate the specificity of MCE method. The results indicated MCE method was capable of specifically detecting high-risk HPV16 and HPV18, and also enabled simultaneous detection of multiplex samples. This MCE method described here has been successfully applied to HPV detection and displayed excellent reliability demonstrating by sequencing results. The inherent capability of MCE facilitated HPV detection conducted in a small chip with automated, high throughput, massive parallelized analysis. We envision that MCE method will definitely pave a way for clinical diagnosis, and even on-site screening of cervical cancer.
Keywords: DNA analysis; Detection; Human papillomavirus; Microchip electrophoresis; Polymerase chain reaction.
© 2020 Xi'an Jiaotong University. Production and hosting by Elsevier B.V.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures





Similar articles
-
[Research progress on analysis of human papillomavirus by microchip capillary electrophoresis].Se Pu. 2020 Oct 8;38(10):1179-1188. doi: 10.3724/SP.J.1123.2020.05016. Se Pu. 2020. PMID: 34213114 Review. Chinese.
-
Visual detection of high-risk HPV16 and HPV18 based on loop-mediated isothermal amplification.Talanta. 2020 Sep 1;217:121015. doi: 10.1016/j.talanta.2020.121015. Epub 2020 Apr 16. Talanta. 2020. PMID: 32498911
-
A novel multiplex real-time PCR assay for the detection and quantification of HPV16/18 and HSV1/2 in cervical cancer screening.Mol Cell Probes. 2012 Apr;26(2):66-72. doi: 10.1016/j.mcp.2012.01.003. Epub 2012 Jan 25. Mol Cell Probes. 2012. PMID: 22293505
-
WarmStart colorimetric LAMP for the specific and rapid detection of HPV16 and HPV18 DNA.J Virol Methods. 2019 Aug;270:87-94. doi: 10.1016/j.jviromet.2019.04.023. Epub 2019 Apr 28. J Virol Methods. 2019. PMID: 31042552
-
Human papillomavirus type 18: association with poor prognosis in early stage cervical cancer.J Natl Cancer Inst. 1996 Oct 2;88(19):1361-8. doi: 10.1093/jnci/88.19.1361. J Natl Cancer Inst. 1996. PMID: 8827013 Review.
Cited by
-
Biosensors for waterborne virus detection: Challenges and strategies.J Pharm Anal. 2023 Nov;13(11):1252-1268. doi: 10.1016/j.jpha.2023.08.020. Epub 2023 Aug 31. J Pharm Anal. 2023. PMID: 38174120 Free PMC article. Review.
-
A Rapid, Sensitive and Type-Specific Detection of High-Risk HPV-16 and HPV-18.J Obstet Gynaecol India. 2023 Oct;73(5):440-444. doi: 10.1007/s13224-023-01751-1. Epub 2023 Apr 19. J Obstet Gynaecol India. 2023. PMID: 37916056 Free PMC article.
-
Recent advancements in nucleic acid detection with microfluidic chip for molecular diagnostics.Trends Analyt Chem. 2023 Jan;158:116871. doi: 10.1016/j.trac.2022.116871. Epub 2022 Dec 5. Trends Analyt Chem. 2023. PMID: 36506265 Free PMC article. Review.
References
-
- Woodman C.B.J., Collins S.I., Young L.S. The natural history of cervical HPV infection: unresolved issues. Nat. Rev. Canc. 2007;7:11–22. - PubMed
-
- Walboomers J.M., Jacobs M.V., Manos M.M. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 1999;189:12–19. - PubMed
-
- Zur Hausen H. Papillomaviruses and cancer: from basic studies to clinical application. Nat. Rev. Canc. 2002;2:342–350. - PubMed
LinkOut - more resources
Full Text Sources

