The Arabidopsis PHD-finger protein EDM2 has multiple roles in balancing NLR immune receptor gene expression
- PMID: 32925902
- PMCID: PMC7529245
- DOI: 10.1371/journal.pgen.1008993
The Arabidopsis PHD-finger protein EDM2 has multiple roles in balancing NLR immune receptor gene expression
Abstract
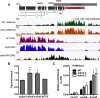
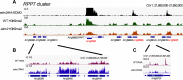
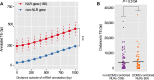
Plant NLR-type receptors serve as sensitive triggers of host immunity. Their expression has to be well-balanced, due to their interference with various cellular processes and dose-dependency of their defense-inducing activity. A genetic "arms race" with fast-evolving pathogenic microbes requires plants to constantly innovate their NLR repertoires. We previously showed that insertion of the COPIA-R7 retrotransposon into RPP7 co-opted the epigenetic transposon silencing signal H3K9me2 to a new function promoting expression of this Arabidopsis thaliana NLR gene. Recruitment of the histone binding protein EDM2 to COPIA-R7-associated H3K9me2 is required for optimal expression of RPP7. By profiling of genome-wide effects of EDM2, we now uncovered additional examples illustrating effects of transposons on NLR gene expression, strongly suggesting that these mobile elements can play critical roles in the rapid evolution of plant NLR genes by providing the "raw material" for gene expression mechanisms. We further found EDM2 to have a global role in NLR expression control. Besides serving as a positive regulator of RPP7 and a small number of other NLR genes, EDM2 acts as a suppressor of a multitude of additional NLR genes. We speculate that the dual functionality of EDM2 in NLR expression control arose from the need to compensate for fitness penalties caused by high expression of some NLR genes by suppression of others. Moreover, we are providing new insights into functional relationships of EDM2 with its interaction partner, the RNA binding protein EDM3/AIPP1, and its target gene IBM1, encoding an H3K9-demethylase.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
The Arabidopsis RRM domain protein EDM3 mediates race-specific disease resistance by controlling H3K9me2-dependent alternative polyadenylation of RPP7 immune receptor transcripts.Plant J. 2019 Feb;97(4):646-660. doi: 10.1111/tpj.14148. Epub 2018 Dec 18. Plant J. 2019. PMID: 30407670 Free PMC article.
-
The PHD-finger module of the Arabidopsis thaliana defense regulator EDM2 can recognize triply modified histone H3 peptides.Plant Signal Behav. 2014;9(7):e29202. doi: 10.4161/psb.29202. Plant Signal Behav. 2014. PMID: 25763495 Free PMC article.
-
An alternative polyadenylation mechanism coopted to the Arabidopsis RPP7 gene through intronic retrotransposon domestication.Proc Natl Acad Sci U S A. 2013 Sep 10;110(37):E3535-43. doi: 10.1073/pnas.1312545110. Epub 2013 Aug 12. Proc Natl Acad Sci U S A. 2013. PMID: 23940361 Free PMC article.
-
Transcript-level expression control of plant NLR genes.Mol Plant Pathol. 2018 May;19(5):1267-1281. doi: 10.1111/mpp.12607. Epub 2017 Nov 20. Mol Plant Pathol. 2018. PMID: 28834153 Free PMC article. Review.
-
Epigenetic gene regulation by plant Jumonji group of histone demethylase.Biochim Biophys Acta. 2011 Aug;1809(8):421-6. doi: 10.1016/j.bbagrm.2011.03.004. Epub 2011 Mar 16. Biochim Biophys Acta. 2011. PMID: 21419882 Review.
Cited by
-
Coordinated Epigenetic Regulation in Plants: A Potent Managerial Tool to Conquer Biotic Stress.Front Plant Sci. 2022 Jan 3;12:795274. doi: 10.3389/fpls.2021.795274. eCollection 2021. Front Plant Sci. 2022. PMID: 35046981 Free PMC article. Review.
-
Immune priming in plants: from the onset to transgenerational maintenance.Essays Biochem. 2022 Sep 30;66(5):635-646. doi: 10.1042/EBC20210082. Essays Biochem. 2022. PMID: 35822618 Free PMC article. Review.
-
Histone H1 prevents non-CG methylation-mediated small RNA biogenesis in Arabidopsis heterochromatin.Elife. 2021 Dec 1;10:e72676. doi: 10.7554/eLife.72676. Elife. 2021. PMID: 34850679 Free PMC article.
-
Widespread premature transcription termination of Arabidopsis thaliana NLR genes by the spen protein FPA.Elife. 2021 Apr 27;10:e65537. doi: 10.7554/eLife.65537. Elife. 2021. PMID: 33904405 Free PMC article.
-
Growth deficiency and enhanced basal immunity in Arabidopsis thaliana mutants of EDM2, EDM3 and IBM2 are genetically interlinked.PLoS One. 2024 Feb 8;19(2):e0291705. doi: 10.1371/journal.pone.0291705. eCollection 2024. PLoS One. 2024. PMID: 38329997 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

