The Influence of Printing Parameters and Cell Density on Bioink Printing Outcomes
- PMID: 32928068
- PMCID: PMC7780841
- DOI: 10.1089/ten.TEA.2020.0210
The Influence of Printing Parameters and Cell Density on Bioink Printing Outcomes
Abstract
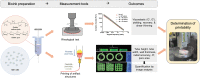
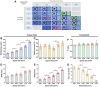
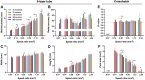
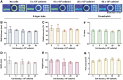
Bioink printability persists as a limiting factor toward many bioprinting applications. Printing parameter selection is largely user-dependent, and the effect of cell density on printability has not been thoroughly investigated. Recently, methods have been developed to give greater insight into printing outcomes. This study aims to further advance those methods and apply them to study the effect of printing parameters (feedrate and flowrate) and cell density on printability. Two printed structures, a crosshatch and five-layer tube, were established as printing standards and utilized to determine the printing outcomes. Acellular bioinks were printed using a testing matrix of feedrates of 37.5, 75, 150, 300, and 600 mm/min and flowrates of 21, 42, 84, 168, and 336 mm3/min. Structures were also printed with cell densities of 5, 10, 20, and 40 × 106 cell/mL at 150 mm/min and 84 mm3/min. Only speed ratios (defined as flowrate divided by feedrate) from 0.07 to 2.24 mm2 were suitable for analysis. Increasing speed ratio dramatically increased the height, width, and wall thickness of tubular structures, but did not influence radial accuracy. For crosshatch structures, the area of pores and the frequency of broken filaments were decreased without impacting pore shape (Pr). Within speed ratios, feedrate and flowrate had negligible, inconsistent effects. Cell density did not affect any printing outcomes despite slight rheological changes. Printing outcomes were dominated by the speed ratio, with feedrate, flowrate, and cell density having little impact on printing outcomes when controlling for speed ratio within the ranges tested. The relevance of these results to other bioinks and printing conditions requires continued investigation by the bioprinting community, as well as highlight speed ratio as a key variable to report and suggest that rheology is a more sensitive measure than printing outcomes.
Keywords: bioink; bioprinting; cell density; federate; flowrate; hydrogel; printability.
Conflict of interest statement
No competing financial interests exist.
Figures
Similar articles
-
Effect of bioink properties on printability and cell viability for 3D bioplotting of embryonic stem cells.Biofabrication. 2016 Sep 16;8(3):035020. doi: 10.1088/1758-5090/8/3/035020. Biofabrication. 2016. PMID: 27634915
-
The influence of printing parameters on cell survival rate and printability in microextrusion-based 3D cell printing technology.Biofabrication. 2015 Nov 2;7(4):045002. doi: 10.1088/1758-5090/7/4/045002. Biofabrication. 2015. PMID: 26523399
-
Simulations of 3D bioprinting: predicting bioprintability of nanofibrillar inks.Biofabrication. 2018 Jun 18;10(3):034105. doi: 10.1088/1758-5090/aac872. Biofabrication. 2018. PMID: 29809162
-
Advancing bioinks for 3D bioprinting using reactive fillers: A review.Acta Biomater. 2020 Sep 1;113:1-22. doi: 10.1016/j.actbio.2020.06.040. Epub 2020 Jul 2. Acta Biomater. 2020. PMID: 32622053 Review.
-
ECM Based Bioink for Tissue Mimetic 3D Bioprinting.Adv Exp Med Biol. 2018;1064:335-353. doi: 10.1007/978-981-13-0445-3_20. Adv Exp Med Biol. 2018. PMID: 30471042 Review.
Cited by
-
Plasma-Based Bioinks for Extrusion Bioprinting of Advanced Dressings.Biomedicines. 2021 Aug 16;9(8):1023. doi: 10.3390/biomedicines9081023. Biomedicines. 2021. PMID: 34440227 Free PMC article.
-
Research Progress in Enzymatically Cross-Linked Hydrogels as Injectable Systems for Bioprinting and Tissue Engineering.Gels. 2023 Mar 15;9(3):230. doi: 10.3390/gels9030230. Gels. 2023. PMID: 36975679 Free PMC article. Review.
-
3D Bioprinting for Engineered Tissue Constructs and Patient-Specific Models: Current Progress and Prospects in Clinical Applications.Adv Mater. 2024 Dec;36(49):e2408032. doi: 10.1002/adma.202408032. Epub 2024 Oct 17. Adv Mater. 2024. PMID: 39420757 Review.
-
3D Bioprinted Head and Neck Squamous Cell Carcinoma (HNSCC) Model Using Tunicate Derived Nanocellulose (NC) Bioink.Adv Healthc Mater. 2025 Mar;14(7):e2403114. doi: 10.1002/adhm.202403114. Epub 2025 Jan 13. Adv Healthc Mater. 2025. PMID: 39801216 Free PMC article.
-
Advanced 3D-Printing Bioinks for Articular Cartilage Repair.Int J Bioprint. 2022 Apr 22;8(3):511. doi: 10.18063/ijb.v8i3.511. eCollection 2022. Int J Bioprint. 2022. PMID: 36105138 Free PMC article. Review.
References
-
- Kang H.W., Lee S.J., Ko I.K., Kengla C., Yoo J.J., and Atala A.. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol 34, 312, 2016 - PubMed
-
- Malda J., Visser J., Melchels F.P., et al. . 25th anniversary article: engineering hydrogels for biofabrication. Adv Mater 25, 5011, 2013 - PubMed
-
- Ozbolat I.T., and Hospodiuk M.. Current advances and future perspectives in extrusion-based bioprinting. Biomaterials 76, 321, 2016 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

