The BB0345 Hypothetical Protein of Borrelia burgdorferi Is Essential for Mammalian Infection
- PMID: 32928963
- PMCID: PMC7671889
- DOI: 10.1128/IAI.00472-20
The BB0345 Hypothetical Protein of Borrelia burgdorferi Is Essential for Mammalian Infection
Abstract
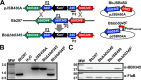
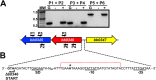
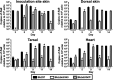
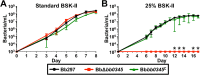
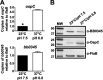
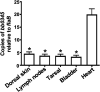
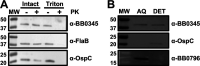
During the natural enzootic life cycle of Borrelia burgdorferi (also known as Borreliella burgdorferi), the bacteria must sense conditions within the vertebrate and arthropod and appropriately regulate expression of genes necessary to persist within these distinct environments. bb0345 of B. burgdorferi encodes a hypothetical protein of unknown function that is predicted to contain an N-terminal helix-turn-helix (HTH) domain. Because HTH domains can mediate protein-DNA interactions, we hypothesized that BB0345 might represent a previously unidentified borrelial transcriptional regulator with the ability to regulate events critical for the B. burgdorferi enzootic cycle. To study the role of BB0345 within mammals, we generated a bb0345 mutant and assessed its virulence potential in immunocompetent mice. The bb0345 mutant was able to initiate localized infection and disseminate to distal tissues but was cleared from all sites by 14 days postinfection. In vitro growth curve analyses revealed that the bb0345 mutant grew similar to wild-type bacteria in standard Barbour-Stoenner-Kelley II (BSK-II) medium; however, the mutant was not able to grow in dilute BSK-II medium or dialysis membrane chambers (DMCs) implanted in rats. Proteinase K accessibility assays and whole-cell partitioning indicated that BB0345 was intracellular and partially membrane associated. Comparison of protein production profiles between the wild-type parent and the bb0345 mutant revealed no major differences, suggesting BB0345 may not be a global transcriptional regulator. Taken together, these data show that BB0345 is essential for B. burgdorferi survival in the mammalian host, potentially by aiding the spirochete with a physiological function that is required by the bacterium during infection.
Keywords: Borrelia; Borrelia burgdorferi; Borreliella; Lyme disease; molecular genetics; pathogenesis; spirochetes.
Copyright © 2020 American Society for Microbiology.
Figures


Similar articles
-
DksA Controls the Response of the Lyme Disease Spirochete Borrelia burgdorferi to Starvation.J Bacteriol. 2019 Jan 28;201(4):e00582-18. doi: 10.1128/JB.00582-18. Print 2019 Feb 15. J Bacteriol. 2019. PMID: 30478087 Free PMC article.
-
Two Distinct Mechanisms Govern RpoS-Mediated Repression of Tick-Phase Genes during Mammalian Host Adaptation by Borrelia burgdorferi, the Lyme Disease Spirochete.mBio. 2017 Aug 22;8(4):e01204-17. doi: 10.1128/mBio.01204-17. mBio. 2017. PMID: 28830947 Free PMC article.
-
Comprehensive Spatial Analysis of the Borrelia burgdorferi Lipoproteome Reveals a Compartmentalization Bias toward the Bacterial Surface.J Bacteriol. 2017 Feb 28;199(6):e00658-16. doi: 10.1128/JB.00658-16. Print 2017 Mar 15. J Bacteriol. 2017. PMID: 28069820 Free PMC article.
-
Regulation of Gene and Protein Expression in the Lyme Disease Spirochete.Curr Top Microbiol Immunol. 2018;415:83-112. doi: 10.1007/82_2017_49. Curr Top Microbiol Immunol. 2018. PMID: 29064060 Review.
-
Borrelia burgdorferi complement regulator-acquiring surface proteins (BbCRASPs): Expression patterns during the mammal-tick infection cycle.Int J Med Microbiol. 2008 Sep 1;298 Suppl 1(Suppl 1):249-56. doi: 10.1016/j.ijmm.2007.10.002. Epub 2007 Dec 31. Int J Med Microbiol. 2008. PMID: 18165150 Free PMC article. Review.
Cited by
-
The Diadenylate Cyclase CdaA Is Critical for Borrelia turicatae Virulence and Physiology.Infect Immun. 2021 May 17;89(6):e00787-20. doi: 10.1128/IAI.00787-20. Print 2021 May 17. Infect Immun. 2021. PMID: 33846120 Free PMC article.
-
InvL, an Invasin-Like Adhesin, Is a Type II Secretion System Substrate Required for Acinetobacter baumannii Uropathogenesis.mBio. 2022 Jun 28;13(3):e0025822. doi: 10.1128/mbio.00258-22. Epub 2022 May 31. mBio. 2022. PMID: 35638734 Free PMC article.
-
Assessment of the hypothetical protein BB0616 in the murine infection of Borrelia burgdorferi.Infect Immun. 2024 Jun 11;92(6):e0009024. doi: 10.1128/iai.00090-24. Epub 2024 May 3. Infect Immun. 2024. PMID: 38700336 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

