Targeting an autocrine IL-6-SPINK1 signaling axis to suppress metastatic spread in ovarian clear cell carcinoma
- PMID: 32929152
- PMCID: PMC7572712
- DOI: 10.1038/s41388-020-01451-4
Targeting an autocrine IL-6-SPINK1 signaling axis to suppress metastatic spread in ovarian clear cell carcinoma
Abstract
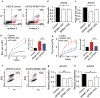
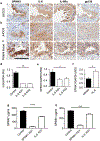
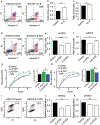
A major clinical challenge of ovarian cancer is the development of malignant ascites accompanied by widespread peritoneal metastasis. In ovarian clear cell carcinoma (OCCC), a challenging subtype of ovarian cancer, this problem is compounded by near-universal primary chemoresistance; patients with advanced stage OCCC thus lack effective therapies and face extremely poor survival rates. Here we show that tumor-cell-expressed serine protease inhibitor Kazal type 1 (SPINK1) is a key driver of OCCC progression and metastasis. Using cell culture models of human OCCC, we find that shRNA silencing of SPINK1 sensitizes tumor cells to anoikis and inhibits proliferation. Knockdown of SPINK1 in OCCC cells also profoundly suppresses peritoneal metastasis in mouse implantation models of human OCCC. We next identify a novel autocrine signaling axis in OCCC cells whereby tumor-cell-produced interleukin-6 (IL-6) regulates SPINK1 expression to stimulate a common protumorigenic gene expression pattern leading to anoikis resistance and proliferation of OCCC cells. We further demonstrate that this signaling pathway can be successfully interrupted with the IL-6Rα inhibitor tocilizumab, sensitizing cells to anoikis in vitro and reducing metastasis in vivo. These results suggest that clinical trials of IL-6 pathway inhibitors in OCCC may be warranted, and that SPINK1 might offer a candidate predictive biomarker in this population.
Conflict of interest statement
Competing Interests
The authors have no competing interests to declare.
Figures



Similar articles
-
Functional Roles of SPINK1 in Cancers.Int J Mol Sci. 2021 Apr 7;22(8):3814. doi: 10.3390/ijms22083814. Int J Mol Sci. 2021. PMID: 33916984 Free PMC article. Review.
-
Targeting STAT3 by HO3867 induces apoptosis in ovarian clear cell carcinoma.Int J Cancer. 2017 Nov 1;141(9):1856-1866. doi: 10.1002/ijc.30847. Epub 2017 Jul 24. Int J Cancer. 2017. PMID: 28646535 Free PMC article.
-
Impact of iASPP on chemoresistance through PLK1 and autophagy in ovarian clear cell carcinoma.Int J Cancer. 2018 Sep 15;143(6):1456-1469. doi: 10.1002/ijc.31535. Epub 2018 May 2. Int J Cancer. 2018. PMID: 29663364
-
Targeting JAK1/STAT3 signaling suppresses tumor progression and metastasis in a peritoneal model of human ovarian cancer.Mol Cancer Ther. 2014 Dec;13(12):3037-48. doi: 10.1158/1535-7163.MCT-14-0077. Epub 2014 Oct 15. Mol Cancer Ther. 2014. PMID: 25319391 Free PMC article.
-
Treatment issues in clear cell carcinoma of the ovary: a different entity?Oncologist. 2006 Nov-Dec;11(10):1089-94. doi: 10.1634/theoncologist.11-10-1089. Oncologist. 2006. PMID: 17110628 Review.
Cited by
-
Serine protease inhibitor Kazal type 1 (SPINK1) promotes proliferation, migration, invasion and radiation resistance in rectal cancer patients receiving concurrent chemoradiotherapy: a potential target for precision medicine.Hum Cell. 2022 Nov;35(6):1912-1927. doi: 10.1007/s13577-022-00776-4. Epub 2022 Sep 2. Hum Cell. 2022. PMID: 36053457 Free PMC article.
-
Inflammatory reprogramming of the solid tumor microenvironment by infiltrating clonal hematopoiesis is associated with adverse outcomes.Cell Rep Med. 2025 Mar 18;6(3):101989. doi: 10.1016/j.xcrm.2025.101989. Epub 2025 Mar 3. Cell Rep Med. 2025. PMID: 40037357 Free PMC article.
-
Functional Roles of SPINK1 in Cancers.Int J Mol Sci. 2021 Apr 7;22(8):3814. doi: 10.3390/ijms22083814. Int J Mol Sci. 2021. PMID: 33916984 Free PMC article. Review.
-
Machine learning and experiments identifies SPINK1 as a candidate diagnostic and prognostic biomarker for hepatocellular carcinoma.Discov Oncol. 2023 Dec 14;14(1):231. doi: 10.1007/s12672-023-00849-2. Discov Oncol. 2023. PMID: 38093163 Free PMC article.
-
Spatially resolved gene expression profiling of tumor microenvironment reveals key steps of lung adenocarcinoma development.Nat Commun. 2024 Dec 6;15(1):10637. doi: 10.1038/s41467-024-54671-7. Nat Commun. 2024. PMID: 39639005 Free PMC article.
References
-
- Howlader N, Noone AM, Krapcho M, Miller D, Brest A, Yu M et al. SEER Cancer Statistics Review, 1975-2016. National Cancer Institute; Bethesda, MD, 2017.
-
- Mitra AK. Ovarian Cancer Metastasis: A Unique Mechanism of Dissemination In: Xu K (ed). Tumor Metastasis. InTech: Rijeka, 2016, p Ch. 03.
-
- du Bois A, Lück H- J, Meier W, Adams H- P, Möbus V, Costa S et al. A Randomized Clinical Trial of Cisplatin/Paclitaxel Versus Carboplatin/Paclitaxel as First-Line Treatment of Ovarian Cancer. Journal of the National Cancer Institute 2003; 95: 1320–1329. - PubMed
-
- Sugiyama T, Kamura T, Kigawa J, Terakawa N, Kikuchi Y, Kita T et al. Clinical characteristics of clear cell carcinoma of the ovary: a distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer 2000; 88: 2584–2589. - PubMed
MeSH terms
Substances
Grants and funding
- R21CA177865/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)/International
- R01CA154387/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)/International
- P50CA136393/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)/International
- R21 CA226302/CA/NCI NIH HHS/United States
- R21 CA177865/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

