Metabolic Constrains Rule Metastasis Progression
- PMID: 32932943
- PMCID: PMC7563739
- DOI: 10.3390/cells9092081
Metabolic Constrains Rule Metastasis Progression
Abstract
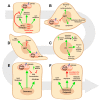
Metastasis formation accounts for the majority of tumor-associated deaths and consists of different steps, each of them being characterized by a distinctive adaptive phenotype of the cancer cells. Metabolic reprogramming represents one of the main adaptive phenotypes exploited by cancer cells during all the main steps of tumor and metastatic progression. In particular, the metabolism of cancer cells evolves profoundly through all the main phases of metastasis formation, namely the metastatic dissemination, the metastatic colonization of distant organs, the metastatic dormancy, and ultimately the outgrowth into macroscopic lesions. However, the metabolic reprogramming of metastasizing cancer cells has only recently become the subject of intense study. From a clinical point of view, the latter steps of the metastatic process are very important, because patients often undergo surgical removal of the primary tumor when cancer cells have already left the primary tumor site, even though distant metastases are not clinically detectable yet. In this scenario, to precisely elucidate if and how metabolic reprogramming drives acquisition of cancer-specific adaptive phenotypes might pave the way to new therapeutic strategies by combining chemotherapy with metabolic drugs for better cancer eradication. In this review we discuss the latest evidence that claim the importance of metabolic adaptation for cancer progression.
Keywords: cancer therapy; epithelial-mesenchymal transition; metastasis; oncometabolism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Menendez-Montes I., Escobar B., Palacios B., Gómez M.J., Izquierdo-Garcia J.L., Flores L., Jimenez-Borreguero L.J., Aragones J., Ruíz-Cabello J., Torres M., et al. Myocardial VHL-HIF Signaling Controls an Embryonic Metabolic Switch Essential for Cardiac Maturation. Dev. Cell. 2016;39:724–739. doi: 10.1016/j.devcel.2016.11.012. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

